Search
Related Product Information
Go to...
Kinases can be classified in different ways: according to sequence homology (as illustrated on the cover) or according to the residue that they phosphorylate: serine or threonine, tyrosine, or a lipid. Tyrosine kinases are further divided into receptor tyrosine kinases that contain an extracellular domain, a transmembrane domain that localizes at the plasma membrane, and an intracellular domain that contains the catalytic domain; or cytoplasmic tyrosine kinases, which are not anchored to the membrane.
Fluorescence resonance energy transfer (FRET) is a distance-dependent interaction between two fluorescent molecules in which energy is transferred from the excited state of a donor molecule to an acceptor molecule, resulting in excitation and fluorescence of the acceptor molecule. The efficiency of FRET is dependent on the inverse sixth power of the intermolecular separation, making it useful over distances comparable with the dimensions of biological macromolecules. Thus, FRET is an important technique for investigating a variety of biological phenomena that produce changes in molecular proximity.
Time-resolved FRET (TR-FRET) utilizes a lanthanide chelate with a long decay lifetime (on the order of milliseconds) as the donor species. This enables detection of acceptor fluorescence after interfering signals from autofluorescent compounds or light scatter from precipitated compounds have decayed (see Figure 1). Therefore TR-FRET is a preferred format for biological assays because of its reduced sensitivity to fluorescent interference.
TR-FRET has additional advantages over traditional FRET in that distance requirements for the interacting components are less stringent. Because of the extended excited-state lifetimes of the Tb or Eu chelate, the macromolecules can undergo conformational changes that bring the donor and acceptor within the correct distance for FRET to occur.
In the LanthaScreen® kinase assays discussed in this module, the donor can be either a Tb or a Eu chelate. The Tb chelate is paired with a fluorescein-labeled acceptor, while the Eu chelate is paired with an Alexa Fluor® 647–labeled acceptor. This module covers two Invitrogen™ LanthaScreen® Assays, LanthaScreen® Kinase Activity Assays and LanthaScreen® Eu Kinase Binding Assays. LanthaScreen® TR-FRET assays are sensitive, typically requiring only nanomolar or subnanomolar amounts of kinase, resulting in cost savings.
 | Figure 1. Schematic of measurement setup for gated detection in a TR-FRET assay. Gated detection of the long lifetime lanthanide chelates such as terbium (Tb) incorporates an initial delay that allows the interfering signal to decay and measurement of the signal due to Tb or Eu chelate at a later time. |
LanthaScreen® Kinase Activity Assays use active kinase, substrate (labeled with acceptor), terbium (Tb)- or europium (Eu)-labeled phosphospecific antibody, and ATP.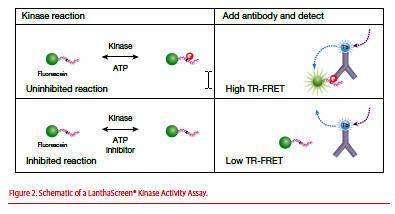
Figure 2. Schematic of a LanthaScreen® Kinase Activity Assay.
LanthaScreen® Activity Assays are typically performed at an ATP concentration equal to the ATP Km[app] for the kinase. The Km is an apparent value because it is only an approximation that is specific for these assay conditions and format. The amount of ATP in the reaction affects the amount of kinase required. The ATP Km values for Invitrogen™ kinases have been determined and are found in the validation packets. The optimal amount of kinase must be determined experimentally by a kinase titration using ATP at its Km.
The optimal amount of kinase is typically the amount required to reach 80% of the maximum signal (EC80) based on a kinase titration curve. The typical starting concentration for such a kinase titration is 100 to 500 ng/mL. Using a concentration of kinase above the EC80 will lead to a slight increase in the assay window but is undesirable as it will underestimate (“right shift”) inhibitor potencies. The IC50 determined for the control inhibitor may then fail the IC50 specification. Using less kinase may reduce the statistical confidence of the assay as measured by the Z’-factor and result in the Z’ failing its specification. However, in some cases, especially with kinases that have low activity, it may be prudent to perform assays at a lower concentration (e.g., EC50) to minimize the amount of kinase necessary and improve the lower limit of IC50 resolution ([kinase]/2).
When using an Invitrogen™ kinase, i is generally only necessary to perform a kinase titration at the predetermined ATP Km prior to performing inhibitor titrations (Steps 3 and 4, respectively, as described in the validation packet). For other sources of kinases, Steps 1 and 2 may need to be performed first to determine the ATP Km. For the purposes of this training, we will assume an Invitrogen™ kinase is being used at its ATP Km.
In general, the kinase titration should cover >5 logs (Figure 3). This can be accomplished by doing a 16-point 2-fold serial dilution of the kinase.
Variation in the activity of a kinase from one manufacturing lot to another may result in differences in the optimal amount of kinase to use. It is generally advisable to redetermine the optimal amount of kinase by kinase titration for any new lots.
The activity assay components needed for each specific kinase can be found in the validation packets.
Note: Optimization steps for a LanthaScreen® Activity Assay as outlined in kinase-specific protocols (validation
packets):
- Kinase titration (EC80 determination) at 1 mM ATP
- ATP Km-apparent determination
- Kinase titration (EC80 determination) at ATP Km
- Inhibitor titration at ATP Km
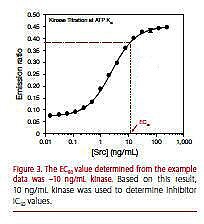
Figure 3. The EC80 value determined from the example data was ~10 ng/mL kinase. Based on this result, 10 ng/mL kinase was used to determine inhibitor IC50 values.
The second category of LanthaScreen® Kinase Assay is a competitive binding assay using an epitope-tagged kinase, a fluorescently labeled ATP competitive “tracer”, and a Eu-labeled anti-epitope tag antibody (Figure 4).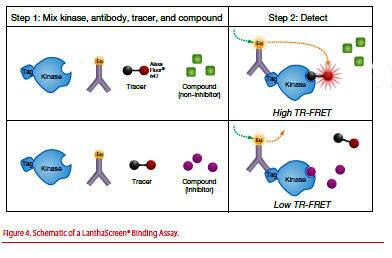
Figure 4. Schematic of a LanthaScreen® Binding Assay.
Assays are typically performed with the tracer concentration close to the dissociation constant (Kd) for the kinase:tracer interaction. For Invitrogen™ kinases, the Kd values have been determined and are in the appropriate detailed protocols. However, the Kd values should be determined experimentally for other sources of kinase.
The binding assay components needed for each specific kinase can be found in the detailed protocols
Go to...
- Previous : General Information
- Next : Practice: Comparison table of materials required
- Frequently asked questions
