Search
Aseptic Laboratory Techniques and Safety in Cell Culture

In a cell culture lab, there are many hazards which require diligent attention to laboratory rules and safety guidelines. In addition, successful cell culture depends heavily on keeping the cells free from contamination by microorganisms such as bacteria, fungi, and viruses. There are proper protocols to handle, discard, and transport cell cultures to help minimize chances of contamination, these are known as aseptic techniques. Aseptic techniques, cell culture lab safety, and personal protective procedures are crucial elements of a safe and high-functioning cell culture lab.
Aseptic laboratory vs. sterile technique
Both aseptic and sterile techniques are essential for maintaining the health of a cell culture lab. The difference between an aseptic technique and sterile technique is that sterile techniques ensure a space is completely free of any microorganisms that could cause contamination, while aseptic techniques focus on not introducing any contamination to a previously sterilized environment. For example, before its first use, a cell culture hood might be sterilized using sterile techniques; while using aseptic techniques will maintain its sterility once a researcher has used it to perform a cell culture experiment.
Read on to learn more about why aseptic techniques are critical to maintaining a sterile work area in your cell culture lab.
What are aseptic laboratory techniques?
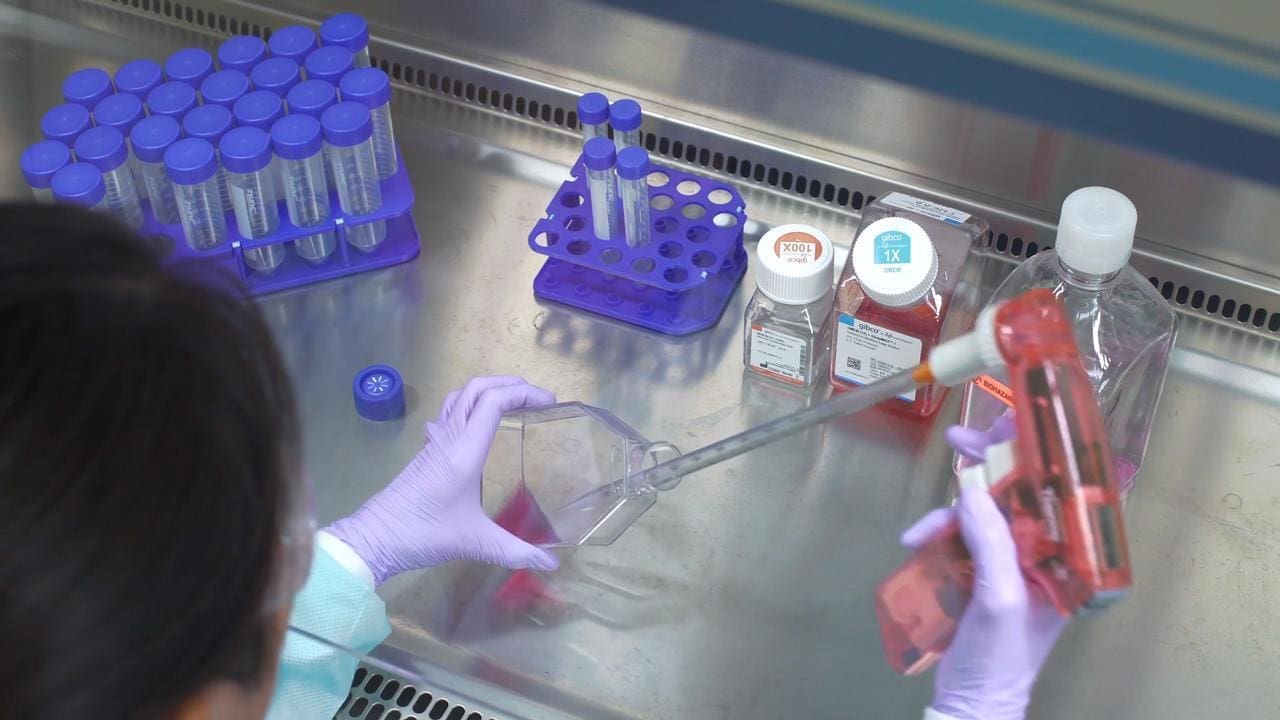
Aseptic technique is a set of procedures designed to create a barrier between microorganisms in the environment and the sterile cell culture, thereby reducing the likelihood of contamination. The primary elements of aseptic laboratory techniques include a sterile work area, good personal hygiene, sterile reagents and media, and sterile handling.
For a list of suggestions and procedures to help you achieve fundamental aseptic laboratory techniques, view our aseptic techniques checklist.
Why is aseptic laboratory technique important?
Aseptic technique in a cell culture lab is important to limit the chance of contamination from biological microorganisms, such as bacteria, fungi, and viruses. Nonsterile supplies, media and reagents, airborne particles laden with microorganisms, unclean incubators, and dirty work surfaces are all sources of biological contamination.
Chemical and biological contaminants, as well as other cell lines can be spread through improper handling and maintenance of your working area. There are many risks when using improper aseptic techniques such as sacrificing the integrity and accuracy of the experiment and wasting valuable lab resources and the time and effort of researchers. You may see altered growth patterns, compromised viability, or even loss of cells. Following a proper aseptic technique, while not 100% fail-safe, gives you a much higher chance of maintaining the health of your cell cultures.
Aseptic laboratory techniques checklist for cell culture
The following checklist provides a concise list of suggestions and procedures to guide you to achieve a solid aseptic laboratory technique. For an in-depth review of aseptic technique, refer to Culture of Animal Cells: A Manual of Basic Technique (Freshney, 2000). For a printable version of the Aseptic techniques checklist for cell culture, request the Cell Culture Basics Handbook.
| Question | Completed | |
|---|---|---|
| Work area | ||
| Is the cell culture hood properly set up? | ||
| Is the cell culture hood in an area free from drafts and through traffic? | ||
| Is the work surface uncluttered, and does it contain only items required for your experiment? | ||
| Did you wipe the work surface with 70% ethanol before work? | ||
| Are you routinely cleaning and sterilizing your incubators, refrigerators, freezers, and other laboratory equipment? | ||
| Personal hygiene | ||
| Did you wash your hands? | ||
| Are you wearing personal protective equipment? | ||
| If you have long hair, is it tied in the back? | ||
| Are you using a pipettor to work with liquids? | ||
| Reagents and media | ||
| Have you sterilized any reagents, media, and solutions you have prepared in the laboratory using the appropriate procedure? | ||
| Did you wipe the outside of the bottles, flasks, and plates with 70% ethanol before placing them on your work surface? | ||
| Are all your bottles, flasks, and other containers capped when not in use? | ||
| Are all your plates stored in sterile re-sealable bags? | ||
| Does any of your reagents look cloudy? Contaminated? Do they contain floating particles? | ||
| Do you detect a foul smell? Unusual color? If yes, did you decontaminate and discard those items? | ||
| Handling | ||
| Are you working slowly and deliberately, mindful of aseptic technique? | ||
| Did you wipe the surfaces of all the items including pipettor, bottles, and flasks with 70% ethanol before placing them in the cell culture hood? | ||
| Are you placing the caps or covers face down on the work area? | ||
| Are you using sterile glass pipettes or sterile disposable plastic pipettes to manipulate all liquids? | ||
| Are you using a sterile pipette only once to avoid cross contamination? | ||
| Are you careful not to touch the pipette tip to anything non-sterile, including the outside edge of the bottle threads? | ||
| Did you mop up any spillage immediately, and wiped the area with 70% ethanol? | ||
Maintaining a sterile work area
A major requirement of a cell culture laboratory is maintaining an aseptic work area that is restricted to cell culture work. Although a separate tissue culture room is preferred, a designated cell culture area within a larger laboratory can still be used for sterile handling, incubation, and storage of cell cultures, reagents, and media. The simplest and most economical way to provide aseptic conditions is to use a laminar flow hood (i.e., biosafety cabinet).
- The cell culture hood should be set up properly in an area with no through traffic. It should be in an area restricted to cell culture and free of drafts from doors, windows, and other equipment.
- The work surface should be uncluttered and contain only items required for a particular procedure; it should not be used as a storage area.
- Before and after use, the work surface should be disinfected thoroughly, and the surrounding areas and equipment should be cleaned routinely.
- For routine cleaning, wipe the work surface with 70% ethanol before and during work, especially after any spillage.
- You may use ultraviolet light to sterilize the air and exposed work surfaces in the cell culture hood between uses.
- Using a Bunsen burner for flaming is not necessary nor is it recommended in a cell culture hood.
- Leave the cell culture hood running at all times, turning it off only when it will not be used for extended periods of time.
Sterile handling
Cell cultures should always be handled with personal protective equipment (PPE). PPE forms an immediate protective barrier between the personnel and the hazardous agent using items such as gloves, laboratory coats and gowns, shoe covers, boots, respirators, face shields, safety glasses, or goggles for protection. Wearing personal protective equipment also helps reduce the probability of contamination from shed skin as well as dirt and dust from your clothes. These items are often used in combination with biosafety cabinets and other devices that contain the agents or materials being handled.
We recommend that you consult your institution’s guidelines for the appropriate use of PPE in your laboratory.
Once dressed in the appropriate PPE, adhering to sterile handling procedures can help protect your cultures, media, and reagents from contamination. Commercial reagents and media undergo strict quality control to help ensure their sterility, but they can become contaminated while handling. Follow the guidelines below for sterile handling to avoid contaminating them. Always sterilize any reagents, media, or solutions prepared in the laboratory using the appropriate sterilization procedure.
- Always wipe your gloved hands and your work area with 70% ethanol.
- Wipe the outside of the containers, flasks, plates, and dishes with 70% ethanol before placing them in the cell culture hood.
- Avoid pouring media and reagents directly from bottles or flasks.
- Use sterile glass or disposable plastic pipettes and a pipettor to work with liquids and use each pipette only once to avoid cross contamination. Do not unwrap sterile pipettes until they are to be used. Keep your pipettes at your work area.
- Always cap the bottles and flasks after use and seal multi-well plates with tape or place them in resealable bags to prevent microorganisms and airborne contaminants from entering.
- Never uncover a sterile flask, bottle, petri dish, etc. until the instant you are ready to use it and never leave it open to the environment. Return the cover as soon as you are finished.
- If you remove a cap or cover and must put it down on the work surface, place the cap with the opening facing down.
- Use only sterile glassware and other equipment.
- Be careful not to talk, sing, or whistle when you are performing sterile procedures.
- Perform your experiments as rapidly as possible to minimize the chance of contamination.
Safe lab practices
Abiding by aseptic techniques in a cell culture lab helps protect the cultures and the people studying them. In addition to the safety risks common to most everyday workplaces, such as electrical and fire hazards, a cell culture laboratory has several specific hazards associated with handling and manipulating human or animal cells and tissues, as well as toxic, corrosive, or mutagenic solvents and reagents. Common hazards are accidental punctures with syringe needles or other contaminated sharps, spills, and splashes onto skin and mucous membranes, ingestion through mouth pipetting, and inhalation exposures to infectious aerosols.
The fundamental objective of any biosafety program is to reduce or eliminate exposure of laboratory workers and the outside environment to potentially harmful biological agents. The most important element of safety in a cell culture laboratory is the strict adherence to standard microbiological practices and techniques.
Guidelines for safe laboratory practices
The following recommendations are simply guidelines for safe lab practices, and they should not be interpreted as a complete code of practice. Consult your institution’s safety committee and follow local rules and regulations pertaining to laboratory safety.
- Refer to your products’ Safety Data Sheet (SDS), also referred to as Material Safety Data Sheet (MSDS) before starting cell culture procedures. The form contains information regarding the properties of a particular substance including physical data such as melting point, boiling and flash point, information on the substance’s toxicity, reactivity, health effects, storage and disposal, as well as recommended protective equipment and procedures for handling spills. The SDSs for all Thermo Fisher Scientific products can be accessed in Documents and Certificates.
- Always wear appropriate personal protective equipment. Change gloves when contaminated and dispose of used gloves with other contaminated laboratory waste.
- Wash your hands before and after working with cell cultures, potentially hazardous materials, and before leaving the laboratory.
- Do not eat, drink, handle contact lenses, apply cosmetics, or store food for human consumption in the laboratory.
- Follow the institutional policies regarding safe handling of sharps (i.e., needles, scalpels, pipettes, and broken glassware).
- Take care to minimize the creation of aerosols and/or splashes.
- Decontaminate all work surfaces before and after your experiments, and immediately after any spill or splash of potentially infectious material with an appropriate disinfectant. Clean laboratory equipment routinely, even if it is not contaminated.
- Decontaminate all potentially infectious materials before disposal.
- Report any incidents that may result in exposure to infectious materials to appropriate personnel (e.g., laboratory supervisor, safety officer).
A companion handbook that introduces the fundamentals of cell culture. Download a copy to keep as a frequent reference guide.
Watch the steps to prevent contamination of your cell culture and a demonstration of best-practice sterile techniques.
Test your cell culture knowledge and refresh on best practices in this virtual cell culture laboratory.
Resources
Cell Culture & Transfection Learning Center
Access cell culture and transfection educational resources to enable excellent experiment planning and execution.
Gibco Cell Culture Basics
Learn best practices and fundamentals of cell culture. Topics include aseptic technique and methods for the growth and maintenance of cells in culture.
Cell Culture Videos
Explore this collection of cell culture videos providing valuable insights into techniques, resources, and stay up to date with the latest advancements in the field.
Cell Culture Selection Tools
Find the right cell culture products for your research with these selection tools. Search for media, FBS, plastics, transfection reagents, and more.
Support
Cell Culture & Transfection Support Center
Find technical support recommendations for your cell culture and transfection workflows, including tips for experimental setup and in-depth troubleshooting help.
Need technical support?
Contact our expert team for technical and application support of Laboratory Products.
Related products
For Research Use Only. Not for use in diagnostic procedures.