Search
Page Contents
The amine-reactive fluorescein derivatives (Amine-reactive xanthene derivatives in this section—Table 1.8) have been the most common fluorescent derivatization reagents for covalently labeling proteins. In addition to its relatively high absorptivity, excellent fluorescence quantum yield and good water solubility, fluorescein (F1300) has an excitation maximum (494 nm) that closely matches the 488 nm spectral line of the argon-ion laser, making it an important fluorophore for confocal laser-scanning microscopy ![]() and flow cytometry applications. In addition, fluorescein's protein conjugates are not inordinately susceptible to precipitation. Because it can be prepared in high purity, fluorescein is one of the five dyes in the Reference Dye Sampler Kit
and flow cytometry applications. In addition, fluorescein's protein conjugates are not inordinately susceptible to precipitation. Because it can be prepared in high purity, fluorescein is one of the five dyes in the Reference Dye Sampler Kit ![]() (R14782, Fluorescence Microscopy Accessories and Reference Standards—Section 23.1). We are also the source of the NIST-traceable fluorescein standard (F36915) described below.
(R14782, Fluorescence Microscopy Accessories and Reference Standards—Section 23.1). We are also the source of the NIST-traceable fluorescein standard (F36915) described below.
NIST-Traceable Fluorescein Standard
The National Institute of Standards and Technology (NIST) chose a high-grade fluorescein synthesized in our laboratories to create Standard Reference Material 1932 (SRM 1932), a certified fluorescein solution. We now offer a NIST-traceable fluorescein standard (F36915) that not only meets the stringent criteria established by NIST, but is also directly traceable to SRM 1932. We supply our NIST-traceable fluorescein standard as a calibrated 50 µM solution of fluorescein in 100 mM sodium borate buffer, pH 9.5; under these conditions, fluorescein is completely ionized ![]() and is therefore in its most fluorescent form (Figure 1.5.1), exhibiting an extremely high quantum yield of 0.93 (Probes Useful at Near-Neutral pH—Section 20.2).
and is therefore in its most fluorescent form (Figure 1.5.1), exhibiting an extremely high quantum yield of 0.93 (Probes Useful at Near-Neutral pH—Section 20.2).
Academic researchers and industry scientists alike can use our NIST-traceable fluorescein standard to assess day-to-day or experiment-to-experiment variation in fluorescence-based instrumentation, as well as to determine the Molecules of Equivalent Soluble Fluorophore (MESF) value for an experimental solution. The MESF value is defined not as the actual number of dye molecules present, but rather as the number of fluorophores that would yield a fluorescence intensity equivalent to that of the experimental solution when analyzed on the same instrument under the same conditions.![]() Consequently, the MESF value is an important tool for characterizing the fluorescence intensity of a solution containing spectrally similar dye molecules attached to antibodies, nucleic acids, microspheres or other substrates that might enhance or diminish the fluorescence. When its pH is carefully matched with that of the experimental solution, our NIST-traceable fluorescein standard can be used for accurate MESF determinations of a wide range of green-fluorescent dye solutions and on an assortment of fluorescence-based instruments.
Consequently, the MESF value is an important tool for characterizing the fluorescence intensity of a solution containing spectrally similar dye molecules attached to antibodies, nucleic acids, microspheres or other substrates that might enhance or diminish the fluorescence. When its pH is carefully matched with that of the experimental solution, our NIST-traceable fluorescein standard can be used for accurate MESF determinations of a wide range of green-fluorescent dye solutions and on an assortment of fluorescence-based instruments.
Limitations of Fluorescein
Even though fluorescein has been used to derivatize biomolecules for decades, fluorescein-based dyes and their conjugates have several significant drawbacks, including:
- A relatively high rate of photobleaching
 (Figure 1.5.2,
(Figure 1.5.2,  )
) - pH-sensitive fluorescence
 (pKa ~6.4) that is significantly reduced below pH 7 (Figure 1.5.3)
(pKa ~6.4) that is significantly reduced below pH 7 (Figure 1.5.3) - A relatively broad fluorescence emission spectrum, limiting their utility in some multicolor applications
- A tendency toward quenching of their fluorescence on conjugation to biopolymers, particularly at high degrees of labeling (Figure 1.5.4)
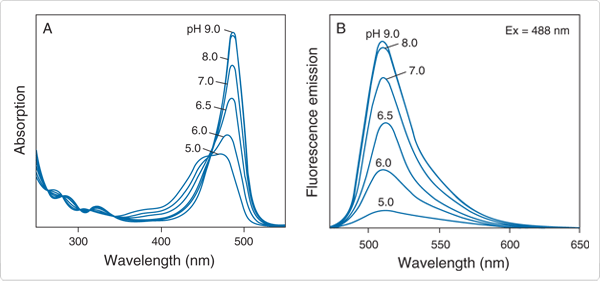
Figure 1.5.1 The pH-dependent spectra of fluorescein (F1300): A) absorption spectra, B) emission spectra.
 |
Figure 1.5.2 Comparison of photostability of green-fluorescent antibody conjugates. The following fluorescent goat anti–mouse IgG antibody conjugates were used to detect mouse anti–human IgG antibody labeling of human anti-nuclear antibodies in HEp-2 cells on prefixed test slides (INOVA Diagnostics Corp.): Oregon Green 514 ( |
 |
Figure 1.5.3 Comparison of pH-dependent fluorescence of the Oregon Green 488 ( |
 |
Figure 1.5.4 Comparison of relative fluorescence as a function of the number of fluorophores attached per protein for goat anti–mouse IgG antibody conjugates prepared using Oregon Green 514 carboxylic acid succinimidyl ester (O6139, |
The photobleaching and pH sensitivity of fluorescein make quantitative measurements with this fluorophore problematic. Furthermore, fluorescein's relatively high photobleaching rate limits the sensitivity that can be obtained, a significant disadvantage for applications requiring ultrasensitive detection, such as DNA sequencing, fluorescence in situ hybridization and localization of low-abundance receptors. These limitations have encouraged the development of alternative fluorophores.
There are no new dyes available that completely solve fluorescein's photobleaching problems, but we have developed some excellent dyes whose spectra mimic those of fluorescein—the Alexa Fluor 488 (Alexa Fluor Dyes Spanning the Visible and Infrared Spectrum—Section 1.3), BODIPY FL (BODIPY Dye Series—Section 1.4), Oregon Green 488, Oregon Green 514 and Rhodamine Green dyes (this section). These dyes are much more photostable than fluorescein and have less or no pH sensitivity in the physiological pH range. When compared with fluorescein, all of these dyes exhibit the same or slightly longer-wavelength spectra (absorption maxima ~490–515 nm) and comparably high fluorescence quantum yields. Alternatively, where they can be used, our yellow-green fluorescent FluoSpheres microspheres and our Qdot nanocrystals (Microspheres—Section 6.5, Qdot Nanocrystals—Section 6.6) provide a means of preparing bioconjugates that have a combination of fluorescence intensity and photostability far superior to that of any simple dye conjugate.
Single-Isomer Fluorescein Isothiocyanate (FITC) Preparations
Despite the availability of alternative amine-reactive fluorescein derivatives that yield conjugates with superior stability and comparable spectra, fluorescein isothiocyanate (FITC) remains one of the most popular fluorescent labeling reagents.![]() The synthesis of fluorescein isothiocyanate, carboxyfluorescein (FAM, see below) and similar fluorescein-derived reagents yields a mixture of isomers at the 5- and 6-positions of fluorescein's carboxyphenyl ring. Spectra of the two isomers are almost indistinguishable in both wavelength and intensity. The isomers, however, may differ in the geometry of their binding to proteins, and the conjugates may elute under different chromatographic conditions or migrate differently in an electrophoretic gel. Thus, certain applications may require the single-isomer preparations. Many fluorescein (and rhodamine) probes are available either as a mixture of isomers or as purified single isomers.
The synthesis of fluorescein isothiocyanate, carboxyfluorescein (FAM, see below) and similar fluorescein-derived reagents yields a mixture of isomers at the 5- and 6-positions of fluorescein's carboxyphenyl ring. Spectra of the two isomers are almost indistinguishable in both wavelength and intensity. The isomers, however, may differ in the geometry of their binding to proteins, and the conjugates may elute under different chromatographic conditions or migrate differently in an electrophoretic gel. Thus, certain applications may require the single-isomer preparations. Many fluorescein (and rhodamine) probes are available either as a mixture of isomers or as purified single isomers.
The 5-isomer or "isomer I" of FITC (F143, ![]() ) is the most widely used FITC isomer, probably because it is easier to isolate in pure form. Because isothiocyanates may deteriorate during storage, we recommend purchasing the 5-isomer of FITC specially packaged in individual vials (F1906, F1907). FITC is readily soluble in aqueous solutions that have a pH above 6. FITC is also available in the FluoReporter FITC Protein Labeling Kit (F6434, Active esters and kits for labeling proteins and nucleic acids—Table 1.2), which is described in Kits for Labeling Proteins and Nucleic Acids—Section 1.2.
) is the most widely used FITC isomer, probably because it is easier to isolate in pure form. Because isothiocyanates may deteriorate during storage, we recommend purchasing the 5-isomer of FITC specially packaged in individual vials (F1906, F1907). FITC is readily soluble in aqueous solutions that have a pH above 6. FITC is also available in the FluoReporter FITC Protein Labeling Kit (F6434, Active esters and kits for labeling proteins and nucleic acids—Table 1.2), which is described in Kits for Labeling Proteins and Nucleic Acids—Section 1.2.
Mixed-Isomer and Single-Isomer Preparations of FAM and JOE Succinimidyl Esters
Although many other companies still prepare their fluorescein bioconjugates with FITC, we prefer to use amine-reactive succinimidyl esters of carboxyfluorescein (commonly called FAM), which yield carboxamides that are more resistant to hydrolysis. We offer single-isomer preparations of FAM (C1359, C1360) and mixed-isomer and single-isomer preparations of FAM succinimidyl esters (C1311, C2210, C6164). A study comparing the relative conjugation rate of several reactive fluorescein derivatives with a protein or L-lysine and the stability of the resulting conjugates concluded that the succinimidyl ester of carboxyfluorescein showed superior performance, followed by fluorescein dichlorotriazine (DTAF, see below). FITC was both the slowest to react and yielded the least stable conjugates;![]() however, the degree of labeling was most easily controlled with FITC.
however, the degree of labeling was most easily controlled with FITC.![]() The succinimidyl ester of 5-FAM (C2210) is reported to react much faster than FITC when used to derivatize small biomolecules prior to separation by capillary electrophoresis.
The succinimidyl ester of 5-FAM (C2210) is reported to react much faster than FITC when used to derivatize small biomolecules prior to separation by capillary electrophoresis.![]() We also offer a single-isomer preparation of the succinimidyl ester of 6-carboxy-4',5'-dichloro-2',7'-dimethoxyfluorescein (6-JOE, SE; C6171MP). 6-JOE is one of the four fluorophores (i.e., 5-FAM, 6-JOE, 6-TAMRA and 6-ROX) used in first-generation electrophoretic DNA sequencing instruments.
We also offer a single-isomer preparation of the succinimidyl ester of 6-carboxy-4',5'-dichloro-2',7'-dimethoxyfluorescein (6-JOE, SE; C6171MP). 6-JOE is one of the four fluorophores (i.e., 5-FAM, 6-JOE, 6-TAMRA and 6-ROX) used in first-generation electrophoretic DNA sequencing instruments.
Succinimidyl Esters of Fluorescein with Spacer Groups
We also prepare succinimidyl esters of fluorescein that contain aliphatic spacers between the fluorophore and the reactive group. These include mixed-isomer (F2181) and single-isomer (F6106) preparations of fluorescein-X succinimidyl ester (SFX), which contains a seven-atom aminohexanoyl spacer ("X") between the FAM fluorophore and the succinimidyl ester. In addition, we offer fluorescein-5-EX succinimidyl ester (F6130), which contains a seven-atom spacer that is somewhat more hydrophilic than is the spacer in SFX. These spacers separate the fluorophore from the biomolecule to which it is conjugated, potentially reducing the quenching that typically occurs upon conjugation. We have determined that conjugates of some proteins prepared with fluorescein-5-EX succinimidyl ester are up to twice as fluorescent as the corresponding conjugates labeled with FITC at the same degree of labeling (Figure 1.5.4). Consequently, we now recommend this fluorescein derivative as the preferred dye for preparing most fluoresceinated proteins. Fluorescein-5-EX succinimidyl ester is also available in our convenient Fluorescein-EX Protein Labeling Kit (F10240). See Kits for Labeling Proteins and Nucleic Acids—Section 1.2 and Molecular Probes kits for protein and nucleic acid labeling—Table 1.3 for more details about these labeling kits.
The spacers in our SFX and fluorescein-5-EX succinimidyl esters may also make the fluorophore more accessible to secondary detection reagents.![]() For example, the spacers should make the fluorescein moiety more available for quenching by our polyclonal and monoclonal anti–fluorescein/Oregon Green antibodies, a technique used to determine the accessibility of the fluorophore in proteins, membranes and cells.
For example, the spacers should make the fluorescein moiety more available for quenching by our polyclonal and monoclonal anti–fluorescein/Oregon Green antibodies, a technique used to determine the accessibility of the fluorophore in proteins, membranes and cells.![]() Fluorescein is frequently used as a hapten on a primary detection reagent that can be either amplified or converted into a longer-wavelength or electron-dense signal with the appropriate secondary detection reagent. Anti-Dye and Anti-Hapten Antibodies—Section 7.4 describes our extensive selection of antibodies to fluorescein and other dyes.
Fluorescein is frequently used as a hapten on a primary detection reagent that can be either amplified or converted into a longer-wavelength or electron-dense signal with the appropriate secondary detection reagent. Anti-Dye and Anti-Hapten Antibodies—Section 7.4 describes our extensive selection of antibodies to fluorescein and other dyes.
Fluorescein Dichlorotriazine (DTAF)
The 5-isomer of fluorescein dichlorotriazine (5-DTAF, D16) is highly reactive with proteins ![]() and is commonly used to prepare biologically active fluorescein tubulin.
and is commonly used to prepare biologically active fluorescein tubulin.![]() Unlike other reactive fluoresceins, 5-DTAF also reacts directly with polysaccharides and other alcohols in aqueous solution at pH above 9, but cannot be used to modify alcohols in the presence of better nucleophiles such as amines or thiols.
Unlike other reactive fluoresceins, 5-DTAF also reacts directly with polysaccharides and other alcohols in aqueous solution at pH above 9, but cannot be used to modify alcohols in the presence of better nucleophiles such as amines or thiols.![]() Polysaccharides that have been modified by DTAF (or other fluorescein derivatives) are readily radioiodinated.
Polysaccharides that have been modified by DTAF (or other fluorescein derivatives) are readily radioiodinated.![]()
Caged Fluorescein Succinimidyl Ester
Caged probes are those that can liberate an active species upon illumination with ultraviolet light (Photoactivatable Reagents, Including Photoreactive Crosslinkers and Caged Probes—Section 5.3). Caged fluorescent dyes can be utilized as polar tracers whose fluorescence can be spatially and temporally activated by illumination. Conjugation of the succinimidyl ester of our water-soluble, caged carboxyfluorescein β-alanine-carboxamide (C20050) to a biomolecule of interest produces an essentially nonfluorescent probe that yields a green-fluorescent fluorescein-labeled product only after ultraviolet photoactivation. Furthermore, caged fluorescein probes are immunochemically cryptic (i.e., the probe is immunoreactive with anti–fluorescein/Oregon Green antibodies after but not before photoactivation). Caged fluorescein succinimidyl ester is also a key starting material for preparing probes for super-resolution photoactivation microscopy.![]()
Oregon Green 488 and Oregon Green 514 dyes are fluorinated analogs of fluoresceins.![]() The absorption and emission spectra of Oregon Green 488 dye (2',7'-difluorofluorescein, D6145) perfectly match those of fluorescein. With additional fluorination of the carboxyphenyl ("bottom") ring of fluorescein, Oregon Green 514 dye exhibits a moderate shift in its absorption and fluorescence spectra of about 15 nm relative to those of fluorescein or Oregon Green 488 dye. Because of the near match of their absorption maxima on proteins (~498 nm and ~512 nm) to the strong 488 nm and 514 nm spectral lines of the argon-ion laser, the Oregon Green 488 and Oregon Green 514 fluorophores are important dyes for both confocal laser-scanning microscopy and flow cytometry applications. Furthermore, spectral imaging systems with linear-unmixing analysis software
The absorption and emission spectra of Oregon Green 488 dye (2',7'-difluorofluorescein, D6145) perfectly match those of fluorescein. With additional fluorination of the carboxyphenyl ("bottom") ring of fluorescein, Oregon Green 514 dye exhibits a moderate shift in its absorption and fluorescence spectra of about 15 nm relative to those of fluorescein or Oregon Green 488 dye. Because of the near match of their absorption maxima on proteins (~498 nm and ~512 nm) to the strong 488 nm and 514 nm spectral lines of the argon-ion laser, the Oregon Green 488 and Oregon Green 514 fluorophores are important dyes for both confocal laser-scanning microscopy and flow cytometry applications. Furthermore, spectral imaging systems with linear-unmixing analysis software ![]() permit the resolution of Oregon Green 514 dye from other green-fluorescent dyes.
permit the resolution of Oregon Green 514 dye from other green-fluorescent dyes.
Bioconjugates prepared from Oregon Green 488 and Oregon Green 514 dyes share several advantages over those of other fluorescein dyes. These include:
- Fluorescence of protein conjugates prepared from Oregon Green 488 and Oregon Green 514 dyes is not appreciably quenched, even at relatively high degrees of labeling (Figure 1.5.4).
- Conjugates of Oregon Green 488 and Oregon Green 514 fluorophores are more photostable than those of fluorescein (Figure 1.5.2, Figure 1.5.5), allowing increased acquisition of photons before photodestruction of the dye and making Oregon Green dyes particularly useful substitutes for fluoresceins for fluorescence imaging applications (
 ).
). - Oregon Green dyes have a lower pKa (pKa = 4.7 versus 6.4 for fluorescein) (Figure 1.5.3), making their fluorescence essentially pH insensitive in the physiological pH range. However, the pH sensitivity of Oregon Green dyes in the weakly acidic range (pH 4 to 6) also makes these dyes useful as pH indicators for acidic organelles of live cells
 (Probes Useful at Acidic pH—Section 20.3).
(Probes Useful at Acidic pH—Section 20.3). - Oregon Green dyes are excellent haptens for anti–fluorescein/Oregon Green antibodies (Anti-Dye and Anti-Hapten Antibodies—Section 7.4, Selected haptenylation reagents and their anti-hapten antibodies—Table 4.2), making Oregon Green bioconjugates useful in a variety of signal amplification schemes.
Both Oregon Green 488 and Oregon Green 514 dyes have also proven useful as fluorescence anisotropy probes for measuring protein–protein and protein–nucleic acid interactions.![]()
 | Figure 1.5.5 Photostability comparison for Oregon Green 514 phalloidin and fluorescein phalloidin (F432). CRE BAG 2 fibroblasts were fixed with formaldehyde, permeabilized with acetone and then stained with the fluorescent phallotoxins. Samples were continuously illuminated and images were acquired every 5 seconds using a Star 1 CCD camera (Photometrics); the average fluorescence intensity in the field of view was calculated with Image-1 software (Universal Imaging Corp.) and expressed as a fraction of the initial intensity. Three data sets, representing different fields of view, were averaged for each labeled phalloidin to obtain the plotted time courses. |
Reactive Oregon Green Dyes
We have prepared a variety of amine-reactive derivatives that enable researchers to take advantage of the spectral properties of Oregon Green 488 and Oregon Green 514 dyes (Amine-reactive xanthene derivatives in this section—Table 1.8). These include the single-isomer succinimidyl esters of Oregon Green 488 carboxylic acid (O6147, O6149) and Oregon Green 514 carboxylic acid (O6139). The 6-isomer of Oregon Green 488-X succinimidyl ester (O6185) contains a seven-atom aminohexanoyl spacer ("X") between the fluorophore and the succinimidyl ester group. This spacer helps to separate the fluorophore from its point of attachment, reducing the interaction of the fluorophore with the biomolecule to which it is conjugated ![]() and making it more accessible to secondary detection reagents.
and making it more accessible to secondary detection reagents.
Oregon Green Protein and Nucleic Acid Labeling Kits
When directly compared with their fluorescein analogs, Oregon Green 488 and Oregon Green 514 conjugates typically have higher fluorescence yields and greater resistance to photobleaching. We have used succinimidyl esters of the Oregon Green 488 and Oregon Green 514 carboxylic acids to prepare conjugates of antibodies (Secondary Immunoreagents—Section 7.2, Summary of Molecular Probes secondary antibody conjugates—Table 7.1), streptavidin (Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6, Molecular Probes avidin, streptavidin, NeutrAvidin and CaptAvidin conjugates—Table 7.9) and a variety of other proteins and ligands. To facilitate direct labeling of biomolecules with Oregon Green dyes, we offer the Oregon Green 488 Protein Labeling Kit (O10241, Kits for Labeling Proteins and Nucleic Acids—Section 1.2).
Carboxyrhodamine 110, which we have named Rhodamine Green dye, is the nonsulfonated analog of Alexa Fluor 488 dye. Rhodamine Green dye offers a combination of desirable properties, including good photostability, a high extinction coefficient (>75,000 cm-1M-1) and a high fluorescence quantum yield, particularly in its nucleotide and nucleic acid conjugates. The Rhodamine Green fluorophore is even more photostable than the Oregon Green 488 dye and about equivalent in photostability to the Oregon Green 514 dye (Figure 1.5.2). Moreover, the fluorescence of its conjugates is completely insensitive to pH between 4 and 9.
Reactive versions of the Rhodamine Green dye (Amine-reactive xanthene derivatives in this section—Table 1.8) were originally developed in our laboratories for use in DNA sequencing and other applications. Rhodamine Green conjugates can be prepared using the amine-reactive succinimidyl ester of Rhodamine Green dye (5(6)-CR 110, SE; R6107) or the succinimidyl ester of the Rhodamine Green-X dye (R6113), which has an additional seven-atom aminohexanoyl spacer ("X") to reduce interaction of the fluorophore and its reaction site. The absorption and fluorescence emission maxima of Rhodamine Green conjugates are red-shifted about 7 nm compared with those of fluorescein; however, they remain compatible with standard fluorescein optical filter sets.
Although the Rhodamine Green dye is one of the most photostable of the fluorescein substitutes, its fluorescence when conjugated to proteins is often substantially quenched, and these conjugates also tend to precipitate from solution. Therefore, we do not recommend any of the Rhodamine Green succinimidyl esters for preparing protein conjugates. However, when conjugated to dextrans,![]() nucleotides and oligonucleotides,
nucleotides and oligonucleotides,![]() the Rhodamine Green fluorophore remains highly fluorescent. In addition, Rhodamine Green dye–labeled probes have been frequently used for fluorescence correlation spectroscopy
the Rhodamine Green fluorophore remains highly fluorescent. In addition, Rhodamine Green dye–labeled probes have been frequently used for fluorescence correlation spectroscopy ![]() (Fluorescence Correlation Spectroscopy (FCS)—Note 1.3).
(Fluorescence Correlation Spectroscopy (FCS)—Note 1.3).
Eosin (2',4',5',7'-tetrabromofluorescein) is usually not chosen for its fluorescence properties—the fluorescence quantum yield is typically only about 10–20% that of fluorescein—but rather for its ability to act as phosphorescent probe or as a fluorescence quencher.![]() With its high quantum yield (~0.57) for singlet oxygen generation, eosin and its conjugates can be used as effective photosensitizers of diaminobenzidine (DAB) oxidation in high-resolution electron microscopy studies (Fluorescent Probes for Photoconversion of Diaminobenzidine Reagents—Note 14.2). The thiol-reactive eosin derivative (eosin-5-maleimide, E118; Thiol-Reactive Probes Excited with Visible Light—Section 2.2) is particularly useful as a phosphorescent probe for measuring the rotational properties of proteins, virus particles and other biomolecules in solution and in membranes.
With its high quantum yield (~0.57) for singlet oxygen generation, eosin and its conjugates can be used as effective photosensitizers of diaminobenzidine (DAB) oxidation in high-resolution electron microscopy studies (Fluorescent Probes for Photoconversion of Diaminobenzidine Reagents—Note 14.2). The thiol-reactive eosin derivative (eosin-5-maleimide, E118; Thiol-Reactive Probes Excited with Visible Light—Section 2.2) is particularly useful as a phosphorescent probe for measuring the rotational properties of proteins, virus particles and other biomolecules in solution and in membranes.![]() In addition, eosin conjugates are employed for fluorescence resonance energy transfer (FRET) studies (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2) and for fluorescence recovery after photobleaching (FRAP) measurements of lateral diffusion.
In addition, eosin conjugates are employed for fluorescence resonance energy transfer (FRET) studies (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2) and for fluorescence recovery after photobleaching (FRAP) measurements of lateral diffusion.
For a detailed explanation of column headings, see Definitions of Data Table Contents
| Cat # | MW | Storage | Soluble | Abs | EC | Em | Solvent | Notes |
|---|---|---|---|---|---|---|---|---|
| C1311 5(6)-FAM, SE | 473.39 | F,D,L | DMF, DMSO | 495 | 74,000 | 519 | pH 9 | 1 |
| 5-FAM | 376.32 | L | pH >6, DMF | 492 | 79,000 | 518 | pH 9 | 1 |
| C1360 6-FAM | 376.32 | L | pH >6, DMF | 492 | 81,000 | 515 | pH 9 | 1 |
| 5(6)-FAM | 376.32 | L | pH >6, DMF | 492 | 78,000 | 517 | pH 9 | 1, 2 |
| C2210 5-FAM, SE | 473.39 | F,D,L | DMF, DMSO | 494 | 78,000 | 520 | pH 9 | 1 |
| 6-FAM, SE | 473.39 | F,D,L | DMF, DMSO | 496 | 83,000 | 516 | pH 9 | 1 |
| C6171MP 6-JOE, SE | 602.34 | F,D,L | DMF, DMSO | 520 | 75,000 | 548 | pH 12 | 3 |
| C20050 CMNB-caged carboxyfluorescein, SE | 962.79 | F,D,LL | DMSO | 289 | 9500 | none | MeOH | 4, 5 |
| D16 5-DTAF | 495.28 | F,D,L | pH >6, DMF | 492 | 83,000 | 516 | pH 9 | 1, 6 |
| D6145 Oregon Green 488 (2',7'-difluorofluorescein) | 368.29 | L | pH >6, DMF | 490 | 87,000 | 514 | pH 9 | 7 |
| eosin-5-ITC | 704.97 | F,DD,L | pH >6, DMF | 521 | 95,000 | 544 | pH 9 | 8, 9 |
| F143 FITC (fluorescein-5-isothiocyanate, Isomer 1) | 389.38 | F,DD,L | pH >6, DMF | 494 | 77,000 | 519 | pH 9 | 1, 8, 10 |
| F1300 fluorescein reference standard | 332.31 | L | pH >6, DMF | 490 | 93,000 | 514 | pH 9 | 1 |
| F1906 FITC | 389.38 | F,DD,L | pH >6, DMF | 494 | 77,000 | 519 | pH 9 | 1, 8, 10 |
| F1907 FITC | 389.38 | F,DD,L | pH >6, DMF | 494 | 77,000 | 519 | pH 9 | 1, 8, 10 |
| F2181 5(6)-SFX | 586.55 | F,D,L | DMF, DMSO | 494 | 74,000 | 520 | pH 9 | 1 |
| F6106 5-SFX | 586.55 | F,D,L | DMF, DMSO | 494 | 75,000 | 521 | pH 9 | 1 |
| F6130 fluorescein-5-EX, SE | 590.56 | F,D,L | DMF, DMSO | 491 | 86,000 | 515 | pH 9 | 1 |
| F36915 fluorescein NIST-traceable standard | 332.31 | RO,L | see Notes | 490 | 93,000 | 514 | pH 9.5 | 1, 11 |
| F2FITC | 425.36 | F,DD,L | DMF, DMSO | 493 | 78,000 | 520 | pH 9 | 7, 8 |
| Oregon Green 514 carboxylic acid | 512.36 | L | pH >6, DMF | 506 | 86,000 | 526 | pH 9 | 12, 13 |
| O6139 Oregon Green 514 carboxylic acid, SE | 609.43 | F,D,L | DMF, DMSO | 506 | 85,000 | 526 | pH 9 | 12, 13 |
| Oregon Green 488 carboxylic acid, 5-isomer | 412.30 | L | pH >6, DMF | 492 | 85,000 | 518 | pH 9 | 7, 14 |
| O6147 Oregon Green 488 carboxylic acid, SE, 5-isomer | 509.38 | F,D,L | DMF, DMSO | 495 | 76,000 | 521 | pH 9 | 7, 14 |
| O6149 Oregon Green 488 carboxylic acid, SE, 6-isomer | 509.38 | F,D,L | DMF, DMSO | 496 | 82,000 | 516 | pH 9 | 7, 14 |
| O6185 Oregon Green 488-X, SE, 6-isomer | 622.53 | F,D,L | DMF, DMSO | 494 | 84,000 | 517 | pH 9 | 7 |
| R6107 Rhodamine Green carboxylic acid, SE (5(6)-CR 110, SE) | 507.89 | F,D,L | DMF, DMSO | 504 | 78,000 | 532 | MeOH | |
| R6113 Rhodamine Green-X, SE | 621.05 | F,D,L | DMF, DMSO | 503 | 74,000 | 528 | MeOH | |
| ||||||||
For Research Use Only. Not for use in diagnostic procedures.