Search
Proteintech
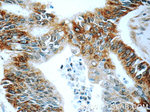
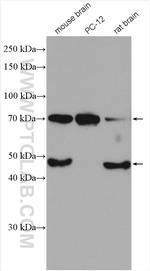
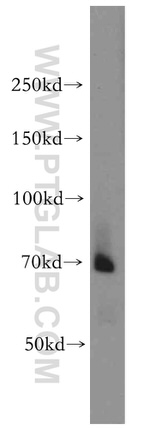
BACE1 Polyclonal Antibody
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
Product Details
12807-1-AP
Species Reactivity
Published species
Host/Isotype
Class
Type
Immunogen
Conjugate
Form
Concentration
Amount
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
Product Specific Information
Immunogen sequence: VRANIAAIT ESDKFFINGS NWEGILGLAY AEIARPDDSL EPFFDSLVKQ THVPNLFSLQ LCGAGFPLNQ SEVLASVGGS MIIGGIDHSL YTGSLWYTPI RREWYYEVII VRVEINGQDL KMDCKEYNYD KSIVDSGTTN LRLPKKVFEA AVKSIKAASS TEKFPDGFWL GEQLVCWQAG TTPWNIFPVI SLYLMGEVTN QSFRITILPQ QYLRPVEDVA TSQDDCYKFA ISQSSTGTVM GAVIMEGFYV VFDRARKRIG FAVSACHVHD EFRTAAVEGP FVTLDMEDCG YNIPQTDEST LM (157-457 aa encoded by BC036084)
Target Information
BACE1 (beta-secretase) is a key enzyme involved in the production of amyloid beta-peptides (Abeta) found in extracellular amyloid plaques of Alzheimer's disease (AD). In some cases early onset familial AD can be attributed to a "Swedish" mutation in the amyloid precursor protein (APP), which dramatically enhances the cleavage of this protein by BACE1. This and other genetic and pathological evidence has led to therapeutic approaches that have focused on the inhibition of BACE1 and other APP-cleaving enzymes, such as gamma-secretase stuff. APP is first cleaved by b-secretase, producing a soluble derivative of the protein and a membrane anchored 99-amino acid carboxy-terminal fragment (C99). The C99 fragment serves as substrate for g-secretase to generate the 4 kDa amyloid-b peptide, which is deposited in the brains of all sufferers of Alzheimer's disease. The long-sought b-secretase was recently identified by several groups independently and designated beta-site APP cleaving enzyme (BACE) and aspartyl protease 2 (Asp2). bACE/Asp2 is a novel transmembrane aspartic protease and colocalizes with APP.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
Bioinformatics
Protein Aliases: APP beta-secretase; asp 2; ASP2; Aspartyl protease 2; BACE; BACE 1; BACE-1B; BACE-1C; BACE-1D; BACE-I-432; BACEdelta7; Beta secretase; Beta-secretase 1; beta-site amyloid beta A4 precursor protein-cleaving enzyme; Beta-site amyloid precursor protein cleaving enzyme 1; Beta-site APP cleaving enzyme 1; beta-site APP processing enzyme; beta-site APP-cleaving enzyme; beta-site APP-cleaving enzyme 1; Memapsin; Memapsin-2; Memapsin2; membrane type aspartyl protease; Membrane-associated aspartic protease 2; transmembrane aspartic proteinase Asp2; unnamed protein product
Gene Aliases: ASP2; BACE; BACE1; C76936; HSPC104; KIAA1149; zgc:77409
UniProt ID: (Human) P56817, (Rat) P56819, (Mouse) P56818
Entrez Gene ID: (Human) 23621, (Rat) 29392, (Mouse) 23821, (Zebrafish) 403005

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support