Search
Invitrogen
CD45 Monoclonal Antibody (HI30), Pacific Orange™
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
FIGURE: 1 / 12
CD45 Antibody (MHCD4530) in Flow












Product Details
MHCD4530
Species Reactivity
Published species
Host/Isotype
Class
Type
Clone
Immunogen
Conjugate
Excitation/Emission Max
Form
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Product Specific Information
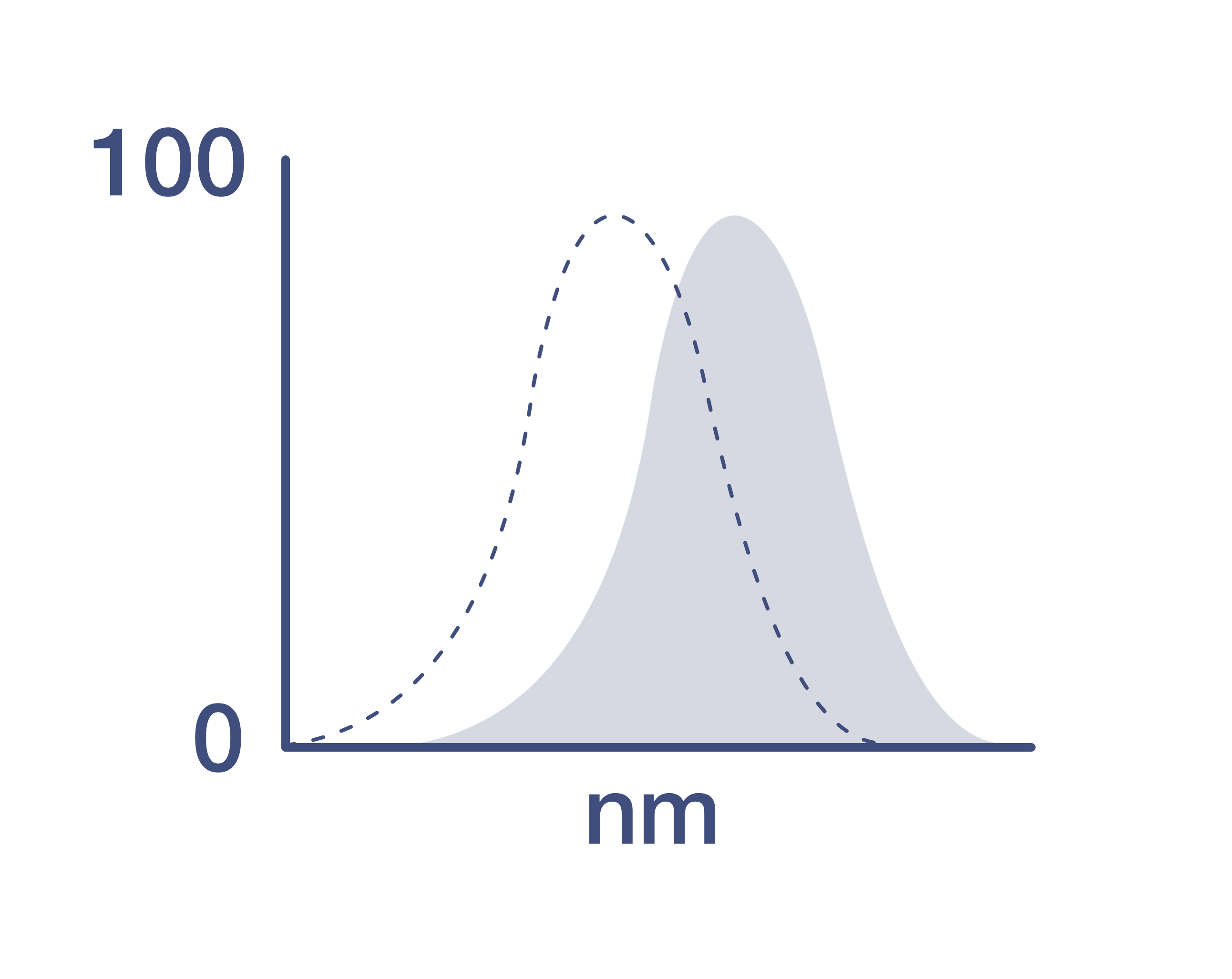
Our Pacific Orange™ dye is optimally excited by the violet laser, and it is recommended that a 575, 585 or 600 nm band pass filter be used for optimal detection. Pacific Blue™ and Pacific Orange™ dye conjugates can be simultaneously excited at 405 nm and emit at 455 nm and 551 nm, respectively, facilitating two-color analysis.
Target Information
CD45 (LCA, leukocyte common antigen) is a receptor-type protein tyrosine phosphatase (PTP) ubiquitously expressed in all nucleated hematopoietic cells, comprising approximately 10% of all surface proteins in lymphocytes. CD45 is absent on non-hematopoietic cell lines, normal and malignant, non-hematopoietic tissues. CD45 glycoprotein is crucial in lymphocyte development and antigen signaling, serving as an important regulator of Src-family kinases. CD45 protein exists as multiple isoforms as a result of alternative splicing, differ in their extracellular domains but share identical transmembrane and cytoplasmic domains. CD45RA is an isoform of the CD45 complex and has restricted expression between different subtypes of lymphoid cells. CD45 isoforms differ in their ability to translocate into the glycosphingolipid-enriched membrane domains and their expression depends on cell type and physiological state of the cell. CD45 has been shown to be an essential regulator of T- and B-cell antigen receptor signaling and suppresses JAK kinases to regulate cytokine receptor signaling. CD45 is also important in promoting cell survival by modulating integrin-mediated signal transduction pathway, DNA fragmentation during apoptosis and inhibition or upregulation of various immunological functions.
Analyte Specific Reagent
How to use the Panel Builder
Watch the video to learn how to use the Invitrogen Flow Cytometry Panel Builder to build your next flow cytometry panel in 5 easy steps.
Bioinformatics
Protein Aliases: CD45; CD45 antigen; glycoprotein 180; L-CA; Leukocyte common antigen; protein tyrosine phosphatase; protein tyrosine phosphatase, receptor type, c polypeptide; Receptor-type tyrosine-protein phosphatase C; T200; T200 glycoprotein; T200 leukocyte common antigen; T220 and B220; unnamed protein product
Gene Aliases: B220; CD45; CD45R; GP180; IMD105; L-CA; LCA; LY5; PTPRC; T200
UniProt ID: (Human) P08575
Entrez Gene ID: (Human) 5788

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support