Search
Invitrogen
CD56 Monoclonal Antibody (MEM-188), PE
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
Product Details
MHCD5604-4
Species Reactivity
Published species
Host/Isotype
Class
Type
Clone
Immunogen
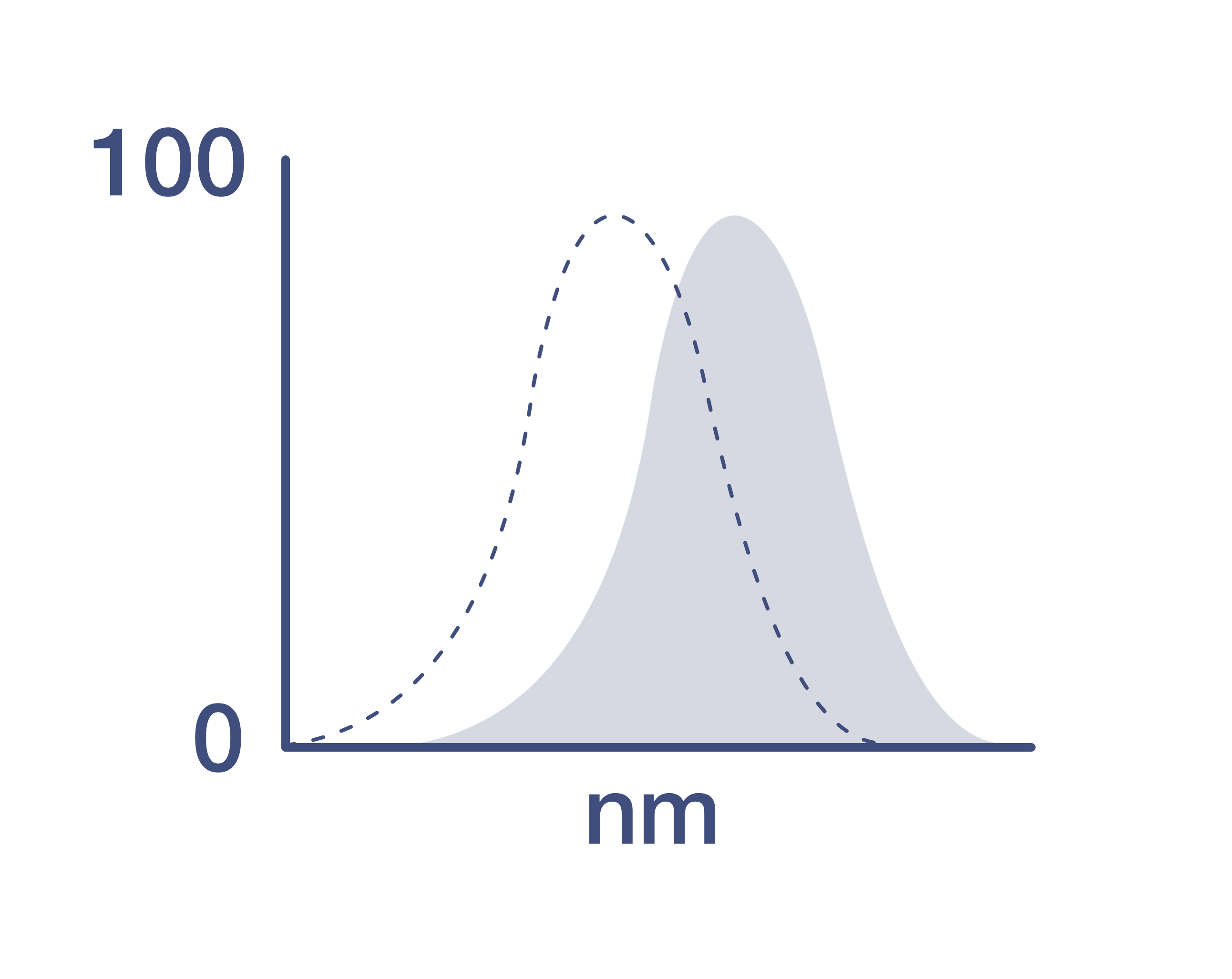
Conjugate
Excitation/Emission Max
Form
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Product Specific Information
R-phycoerythrin (PE) is a stable and highly soluble phycobiliprotein which provides maximal absorbance and fluorescence without susceptibility to internal or external fluorescence quenching, thus providing an exceptional quantum yields and molar extinction coefficients.
Target Information
CD56, also known as neural cell adhesion molecule (NCAM), is a highly glycosylated transmembrane glycoprotein of the immunoglobulin family. It plays a crucial role in cell adhesion, migration, axonal growth, pathfinding, and synaptic plasticity. CD56 is ubiquitously expressed in the nervous system in isoforms ranging from 120-180 kDa and is involved in homotypic adhesion of neural cells. It mediates interactions by binding extracellular matrix components such as laminin and integrins, with polysialic modification reducing CD56-mediated adhesion. In the hematopoietic system, CD56 is expressed on natural killer (NK) cells and a subset of T cells known as NKT cells. It is also found on most neuroectodermal-derived cell lines, tissues, and neoplasms, including retinoblastoma, medulloblastoma, astrocytomas, and neuroblastoma. CD56 serves as a widely used neuroendocrine marker with high sensitivity for neuroendocrine tumors and ovarian granulosa cell tumors. Diseases associated with CD56 dysfunction include rabies and blastic plasmacytoid dendritic cell neoplasms, highlighting its importance in both neural and immune system functions.
Analyte Specific Reagent
How to use the Panel Builder
Watch the video to learn how to use the Invitrogen Flow Cytometry Panel Builder to build your next flow cytometry panel in 5 easy steps.
Bioinformatics
Protein Aliases: 145 kda neural cell adhesion molecule; antigen recognized by monoclonal antibody 5.1H11; CD-56; CD56; E NCAM; membrane glycoprotein; N CAM1; N-CAM; N-CAM-1; neural cell adhesion molecule; Neural cell adhesion molecule 1; neural cell adhesion molecule secreted isoform; neural cell adhesion molecule, NCAM; sCD56; sNCAM; soluble CD56; soluble NCAM; unnamed protein product
Gene Aliases: CD56; MSK39; NCAM; NCAM1
UniProt ID: (Human) P13591
Entrez Gene ID: (Human) 4684

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support