Search
Invitrogen
ERO1L Polyclonal Antibody, DyLight™ 550
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
Product Details
PA5-22746
Host/Isotype
Class
Type
Immunogen
Conjugate
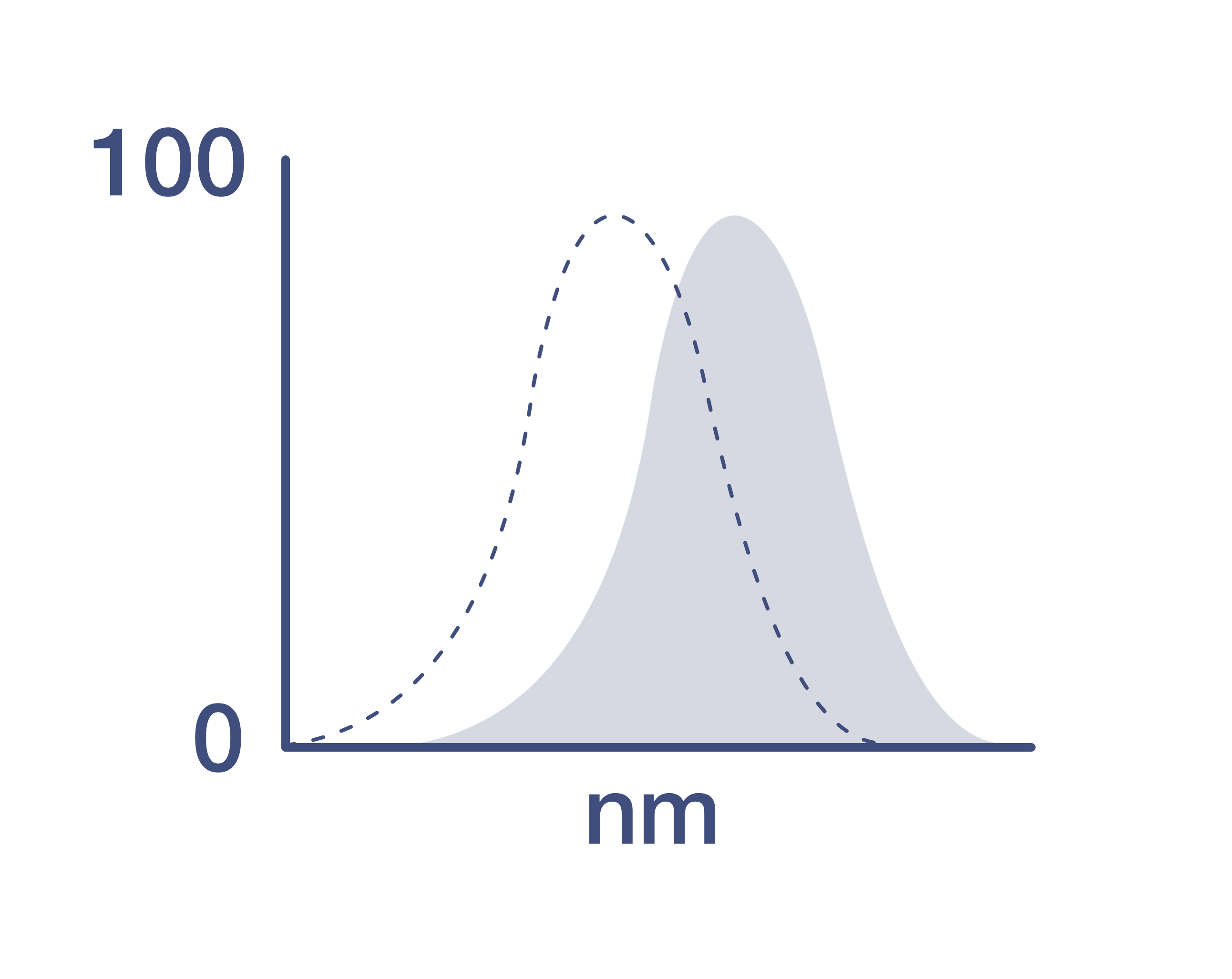
Excitation/Emission Max
Form
Concentration
Amount
Storage conditions
Shipping conditions
RRID
Product Specific Information
This ERO1L antibody is useful for Western Blot, where a band ~57 kDa is observed.
Depending on the samples used ERO1L can also appear as a doublet which is believed to represent the oxidized and reduced forms of the protein.
Suggested positive control: 293T, INS-1, and MIN6 cell lysate.
Target Information
ERO1L (endoplasmic oxidoreductin-1-like protein) is localized in ER membrane as peripheral membrane protein on the lumenal side. Its association with ERP44 is essential for its retention in the ER and is unique in coupling oxygen reduction to de novo disulfides formation. The ERO1L enzyme is broadly distributed at low levels in several tissues with highest levels in the upper digestive tract and is stimulated by hypoxia via HIF-signaling. ERO1L generally exists as monomer, but has the ability to function as monomer or homodimer and interacts with PDILT. After translation, ERO1L gets n-glycosylated and the disulfide bonds constitute the redox-active center whereby the Cys-94/Cys-99 disulfide bond accepts an electron from P4HB followed by funneling the same to the active site disulfide Cys-394/Cys-397. ERO1L acts by oxidizing directly to P4HB/PDI isomerase through direct disulfide exchange and associates with ERP44. ERO1L is implicated in immunoglobulin folding, oxidative stress, release of unfolded cholera toxin from reduced P4HB/PDI upon Vibrio cholerae infection, and plays a key role in ER stress-induced CHOP-dependent apoptosis through inositol 1,4,5-trisphosphate receptor IP3R1 activation.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
References (0)

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support