Search Thermo Fisher Scientific
Invitrogen
Goat anti-Rat IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 647
FIGURE: 1 / 19
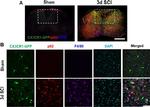
Rat IgG (H+L) Cross-Adsorbed Secondary Antibody (A-21247) in WB














Product Details
A-21247
Western Blot (WB)
Immunohistochemistry (IHC)
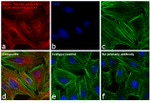
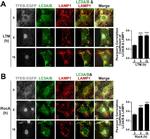
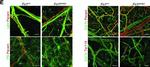
Immunocytochemistry (ICC/IF)
Immunoprecipitation (IP)
Species Reactivity
Host/Isotype
Class
Type
Immunogen
Conjugate
Excitation/Emission Max
Form
Concentration
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Target
Cross Adsorption
Antibody Form
Product Specific Information
To minimize cross-reactivity, these goat anti-rat IgG (H+L) whole secondary antibodies have been affinity purified and cross-adsorbed against mouse IgG, mouse serum, and human serum. Cross-adsorption or pre-adsorption is a purification step to increase specificity of the antibody resulting in higher sensitivity and less background staining. The secondary antibody solution is passed through a column matrix containing immobilized serum proteins from potentially cross-reactive species. Only the nonspecific-binding secondary antibodies are captured in the column, and the highly specific secondaries flow through. The benefits of this extra step are apparent in multiplexing/multicolor-staining experiments (e.g., flow cytometry) where there is potential cross-reactivity with other primary antibodies or in tissue/cell fluorescent staining experiments where there are may be the presence of endogenous immunoglobulins.
Alexa Fluor dyes are among the most trusted fluorescent dyes available today. Invitrogen™ Alexa Fluor 647 dye is a near-infrared-fluorescent dye with excitation ideally suited to the 647 nm laser line. For stable signal generation in imaging and flow cytometry, Alexa Fluor 647 dye is pH-insensitive over a wide molar range. Probes with high fluorescence quantum yield and high photostability allow detection of low-abundance biological structures with great sensitivity. Alexa Fluor 647 dye molecules can be attached to proteins at high molar ratios without significant self-quenching, enabling brighter conjugates and more sensitive detection. The degree of labeling for each conjugate is typically 2-8 fluorophore molecules per IgG molecule; the exact degree of labeling is indicated on the certificate of analysis for each product lot.
Using conjugate solutions: Centrifuge the protein conjugate solution briefly in a microcentrifuge before use; add only the supernatant to the experiment. This step will help eliminate any protein aggregates that may have formed during storage, thereby reducing nonspecific background staining. Because staining protocols vary with application, the appropriate dilution of antibody should be determined empirically. For the fluorophore-labeled antibodies a final concentration of 1-10 µg/mL should be satisfactory for most immunohistochemistry and flow cytometry applications.
Product will be shipped at Room Temperature.
Target Information
Anti-Rat secondary antibodies are affinity-purified antibodies with well-characterized specificity for rat immunoglobulins and are useful in the detection, sorting or purification of its specified target. Secondary antibodies offer increased versatility enabling users to use many detection systems (e.g. HRP, AP, fluorescence). They can also provide greater sensitivity through signal amplification as multiple secondary antibodies can bind to a single primary antibody. Most commonly, secondary antibodies are generated by immunizing the host animal with a pooled population of immunoglobulins from the target species and can be further purified and modified (i.e. immunoaffinity chromatography, antibody fragmentation, label conjugation, etc.) to generate highly specific reagents.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support