Search
Bioss
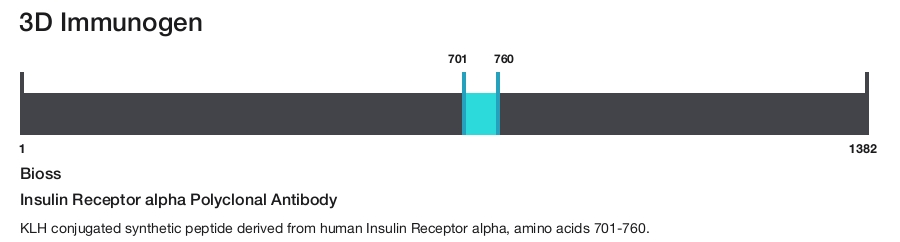
Insulin Receptor alpha Polyclonal Antibody
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
FIGURE: 1 / 5
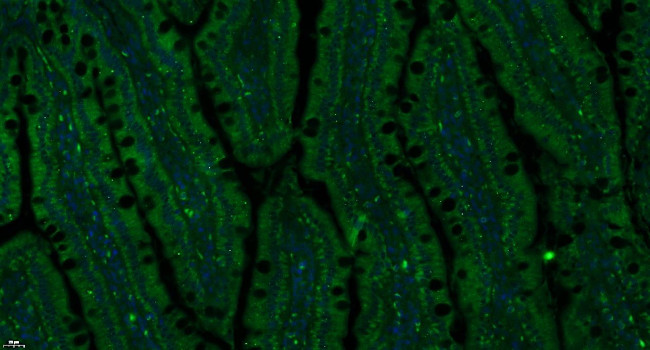
Insulin Receptor alpha Antibody (BS-0047R) in IHC (P)





Product Details
BS-0047R
Species Reactivity
Host/Isotype
Class
Type
Immunogen
Conjugate
Form
Concentration
Amount
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
Target Information
Biological actions of insulin and IGF-1 are mediated by their respective cell surface receptor tyrosine kinases that regulate multiple signaling pathways through activation of a series of phosphorylation cascades. The insulin receptor. Insulin/IGF-1 binding to the extracellular domain leads to autophosphorylation of downstream target proteins. These two receptors differ in sequence in regions that confer specificity for the designated ligand as well as in certain intracellular signaling domains, resulting in significant differences in the functional consequences of activation of each receptor. Defects in IR are the cause of various insulin resistance syndromes and IGF-1R defects may cause some forms of growth retardation. Both these signaling cascades are also important for the development of cancer.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
References (0)
Bioinformatics
Protein Aliases: CD220; CD220 beta; Insulin receptor; insulin receptor precursor; insulin receptor preproprotein; IR; IR alpha; IR beta; unnamed protein product
Gene Aliases: 4932439J01Rik; CD220; D630014A15Rik; HHF5; INSR; IR; IR-A; IR-B
UniProt ID: (Human) P06213, (Mouse) P15208
Entrez Gene ID: (Human) 3643, (Rat) 24954, (Mouse) 16337

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support