Search
Proteintech
RGS14 Polyclonal Antibody
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
Product Details
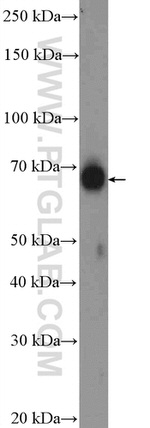
16258-1-AP
Species Reactivity
Published species
Host/Isotype
Class
Type
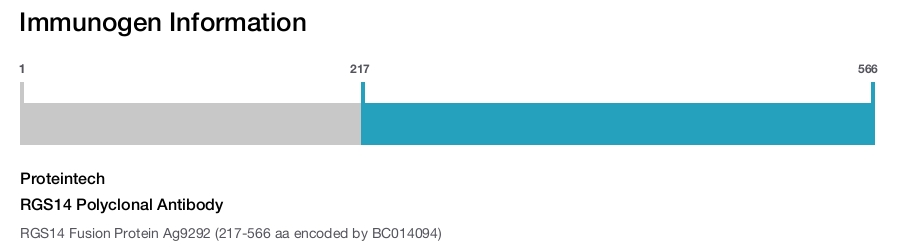
Immunogen
Conjugate
Form
Concentration
Amount
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
Product Specific Information
Immunogen sequence: KSLPLGVEE LGQLPPVEGP GGRPLRKSFR RELGGTANAA LRRESQGSLN SSASLDLGFL AFVSSKSESH RKSLGSTEGE SESRPGKYCC VYLPDGTASL ALARPGLTIR DMLAGICEKR GLSLPDIKVY LVGNEQALVL DQDCTVLADQ EVRLENRITF ELELTALERV VRISAKPTKR LQEALQPILE KHGLSPLEVV LHRPGEKQPL DLGKLVSSVA AQRLVLDTLP GVKISKARDK SPCRSQGCPP RTQDKATHPP PASPSSLVKV PSSATGKRQT CDIEGLVELL NRVQSSGAHD QRGLLRKEDL VLPEFLQLPA QGPSSEETPP QTKSAAQPIG GSLNSTTDSA L (217-566 aa encoded by BC014094)
Target Information
RGS14 is a member of the regulator of G-protein signaling family which contains one RGS domain, two Raf-like Ras-binding domains (RBDs), and one GoLoco domain. RGS14 attenuates the signaling activity of G-proteins by binding, through its GoLoco domain, to specific types of activated, GTP-bound G alpha subunits. Acting as a GTPase activating protein (GAP), the protein increases the rate of conversion of the GTP to GDP. The resulting hydrolysis allows the G alpha subunits to bind G beta/gamma subunit heterodimers, forming inactive G-protein heterotrimers, thereby terminating the signal. Alternate transcriptional splice variants of RGS14 have been observed but have not been thoroughly characterized. Further, RGS14 acts as a positive modulator of microtubule polymerisation and spindle organization through a G(i)-alpha-dependent mechanism; plays a role in cell division; is required for the nerve growth factor (NGF)-mediated neurite outgrowth, and is involved in stress resistance. RGS14 protein may also be involved in visual memory processing capacity and hippocampal-based learning and memory.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
Bioinformatics
Protein Aliases: rap1/rap2 interacting protein; RAP1/RAP2-interacting protein; Regulator of G-protein signaling 14; RGS14; RGSE; RPIP1; unnamed protein product
Gene Aliases: RGS14; RPIP1
UniProt ID: (Human) O43566, (Mouse) P97492
Entrez Gene ID: (Human) 10636, (Mouse) 51791

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support