Quantifiable consistency. By tracking retention time consistency and peak symmetry across years of production, column performance becomes directly comparable.
In chromatography, inconsistent separations are inconvenient and costly.
Every unexpected shift in retention time or change in peak shape can trigger a cascade of troubleshooting, reruns, and uncertainty that slows laboratory productivity.

On the other hand, when columns perform consistently, laboratories recover valuable time, reduce uncertainty, and maintain confidence in their data. True batch-to-batch consistency means fewer failed validations, fewer investigations, easier audits, and smoother collaboration across global sites.
In regulated industries, data integrity underpins every decision, from early development to final release testing, so consistent column performance is a necessity.
Key takeaways
- Batch-to-batch (U)HPLC column consistency directly influences long-term method performance by stabilizing retention times, preserving peak shape, and supporting reliable validation of pharmaceutical methods.
- Multi-year performance data across thousands of columns shows us how controlled column manufacturing reduces chromatographic variability, supporting reproducible results across column formats, batches, and laboratories.
- Stable (U)HPLC columns minimize revalidation, troubleshooting, and method transfer challenges in regulated workflows, improving analytical efficiency across the product lifecycle.
Defining and measuring batch-to-batch (U)HPLC column consistency
A (U)HPLC column is packed with solid-phase particles that separate compounds by chemical structure.
If medium composition differs even slightly between manufacturing batches, retention times and peak shapes can change. These differences might seem minor, but in pharmaceutical analysis they can trigger failed validations, unnecessary troubleshooting, and inconsistent data across labs.
A recent evaluation of Thermo Scientific Hypersil GOLD columns examined whether these columns perform consistently across years of production. The answer: our results found sustained performance over time.
Across roughly 17,500 columns tested over three years, researchers focused on two major indicators of a manufacturer’s column performance:
- Relative retention time (RRT), which is essentially how consistently compounds elute off the column.
- Peak asymmetry, a reflection of column packing quality and how well peaks “behave.”
In a recent technical note: Evaluation of batch-to-batch consistency of reversed-phase HPLC columns for long-term method validation, we evaluated three column configurations: a 250 mm × 4.6 mm, 5 μm column; a 150 mm × 4.6 mm, 5 μm column; and 50 mm × 2.1 mm, 5 μm column, using a test mixture of five compounds selected to represent different hydrophobicities.
Despite differences in column geometry and media batches, the method produced highly consistent chromatographic performance.
The method also achieved RRT relative standard deviation variation of no more than 2.11% for the compounds and kept peak asymmetry within a narrow range of 0.92 to 1.09. This level of control reduces uncertainty from long-term method validation, reinforcing confidence in predictable, reproducible column performance.
Why column stability over time matters
As an experienced analyst, you already know how frustrating method validation can be. You think you’ve nailed the method, only to find the results from run to run. Often, the issue isn’t you. It’s the tools.
When your column behaves the same every time, you will see several downstream benefits, such as:
1. Improved confidence in data interpretation
You don’t have to keep wondering whether differences in your data are coming from your chemistry or your column.
2. Methods stay valid longer
Pharmaceutical companies must monitor drug purity throughout the product lifecycle, from development to manufacturing to quality control. A column that performs consistently over time makes comparisons straightforward and reliable.
3. Method transfer is more reliable
Large companies operate across multiple global sites. When everyone uses columns that behave consistently, transferring methods becomes seamless rather than a months-long headache.
A closer look at column performance data
The published technical note visualizes column performance using clean, simple charts (Figure 1).
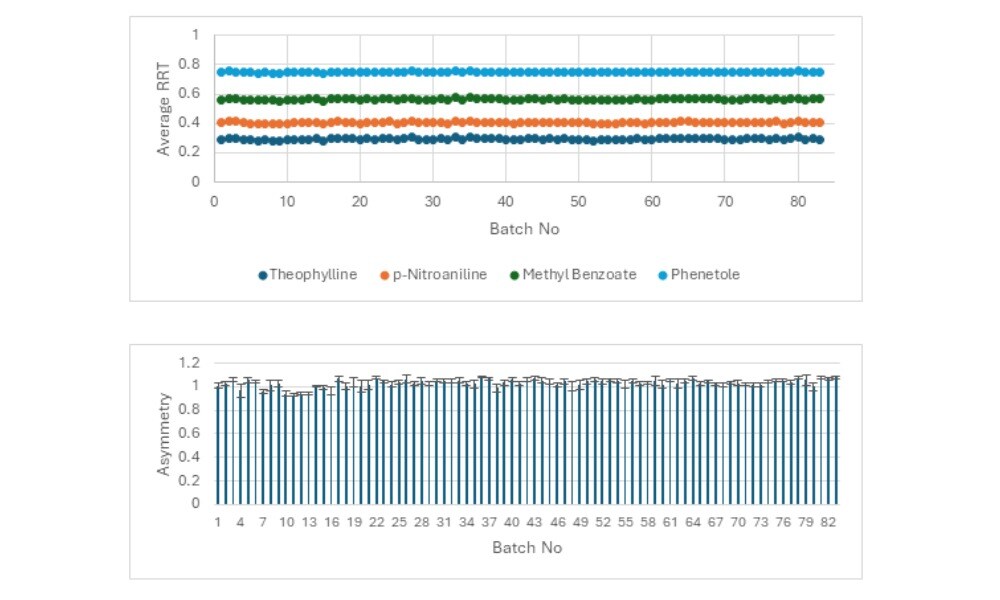
Figure 1. (Top image) Batch-wise average RRT, normalized by o-Xylene for Hypersil GOLDTM 250 mm x 4.6 mm 5 µm (83 batches). (Bottom image) Average peak asymmetry evaluation of o-xylene Hypersil GOLDTM 250 mm x 4.6 mm 5 µm (83 batches).
These graphs show us:
- Tight clusters of RRT points across 83 batches, depending on column format.
- Nearly identical behavior between columns of different dimensions, which is a sign of effective manufacturing process controls.
- Peak asymmetry values hovering around 1.0, indicating well-packed columns producing sharp, reliable peaks.
This data might not seem glamorous at first glance, but for anyone who’s spent hours troubleshooting peak tailing, drifting retention times, or inconsistent quality control (QC) results, these charts are beautiful.
Why column consistency matters for long-term method performance
In automated LC workflows, a small change in column behavior can affect multiple downstream outputs: integration, peak purity assessment, and calculated assay/impurity results. Over time, this can translate into system suitability failures, drift outside validated ranges, and additional validation or comparability work.
Column stability and batch-to-batch reproducibility are therefore critical for sustaining validated methods over time. The multi-batch data presented here demonstrate how controlled manufacturing processes can maintain consistent retention behavior and peak shape over time.
To learn how to test your column and determine what data to look for, read our technical note, Evaluation of batch-to-batch consistency of reversed phase HPLC columns for long-term method validation.
FAQs
Batch-to-batch consistency has a direct impact on the stability of chromatographic results. When columns are manufactured consistently, retention times remain stable and compounds elute at predictable intervals — run after run and batch after batch.
This stability reduces unexpected shifts in selectivity or peak shape, helping analysts distinguish true method-related changes from column-related variability.
Batch-to-batch consistency is evaluated by applying the same standardized test method to columns produced across multiple manufacturing lots and by comparing key chromatographic performance metrics.
Commonly monitored parameters include RRT, which reflects retention stability and peak asymmetry, which indicates packing quality and peak integrity. By tracking these metrics over many batches and extended production periods, manufacturers can quantify variability and ensure controlled, reproducible column performance.
Consistent LC column performance plays an important role in sustaining validated methods over time, particularly in regulated pharmaceutical environments.
When column behavior remains stable across production batches, methods are more likely to stay within established acceptance criteria, reducing the need for revalidation or investigation. This stability is especially valuable throughout the product lifecycle, from development through quality control and release testing.
Batch-to-batch consistency also facilitates smoother method transfer between laboratories and global sites. When different labs use columns that perform the same way, chromatographic results are more predictable, making transfers faster, more efficient, and less prone to variability-related delays.
Visit us on LinkedIn: #UHPLC #ColumnsAndConsumables #pharmaceutical



