
Pulsation in Peristaltic Pumps vs Non-Peristaltic Pumps
First we need to understand what a peristaltic pump is. In peristaltic pumps, fluid is pumped by running rollers over a hose. The rollers result in pulsations that are proportional to the number of rollers and RPM of the pump. As a result, peristaltic pumps generate pulsations in their output just by the very nature of their design. Typically, for this reason, peristaltic pumps are not used for continuous steady fluid delivery.
Recently, several cytometers have come to the market with peristaltic pump designs with dampening systems and claims of “pulsation-less flow”. So, is it possible for a peristaltic-based system to be pulsation-less and why do we care?
Let’s have a quick look to see if there is any fluid pulsation in the peristaltic driven system as compared to an instrument with a non-peristaltic, continuous flow pump. Here we compare some data from a flow cytometer with a peristaltic pump and the Attune™NxT flow cytometer with a non-peristaltic pump.
To observe pulsations, we will plot Time vs. Count. This results in a histogram that gives a moving average of how many events are showing up at a given time.
Figure 1shows an example from a flow cytometer with a peristaltic pump. As can be seen in this figure, the fluid pulsation in the peristaltic pump system is quite large . . . +/- 33% of the average count . . . or a total spread of 66% of the average count!

Figure 1.
Now that we understand the variation in count over time in a flow cytometer with a peristaltic pump, let’s compare this to the Attune NxT flow cytometer with a pump that is designed and engineered to create ‘smooth’ fluid delivery. The same test was performed and the data is shown in Figure 2 below. You can see from the data below that the count is much more stable with a variable of only +/- 5%.
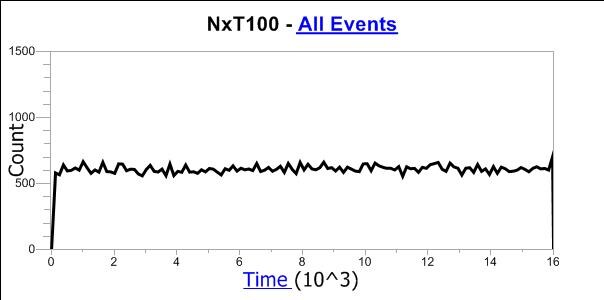
Figure 2
Why Should We Care?
There are several reasons for necessitating stable flow. One of the top reasons is for precise counts. If the count fluctuates rapidly, the precision of the absolute count will also fluctuate rapidly. If one wants reliable counts, then stable fluid delivery cannot be compromised.
This also affects coincidence rate. Pulsations in the flow result in variations in the particle’s velocity within the flow stream. The larger the pulsations, the larger the uncertainty of where the particle will be after traversing a distance within the flow cell. For single laser systems this is not an issue as all particles are analyzed on the laser triggering the experiment. For systems with multiple pinholes, the uncertainty in particle position causes manufacturers to increase the window size of where the particle should be located. The greater the number of pinholes, the greater the uncertainty of particle position in the flow cell.
As an example, consider a system where a particle spends 3 µseconds in the laser. For systems with small variation in the particle velocity and multiple pinholes, the interrogation window may be ~5 µs to account for any uncertainties in particle velocity. If there is a large fluctuation in the fluidic system, the designer may need to increase the interrogation window width to 10 µs or greater to ensure the particle appears within the window. Unfortunately, increasing the interrogation window width also increases the coincidence rate. The probability of having two particles occur in a five µs window is much smaller than the probability of two particles occurring in a ten microsecond window.
As a general rule, in comparing cytometers for coincidence rate, it is the electronic interrogation window that determines coincidence, not the amount of time the particle spends in the laser. Fluidic pulsation is a large contributor to electronic interrogation window width and thus coincidence.
Leave a Reply