Search
Invitrogen
CD8 Monoclonal Antibody (3B5), APC
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
Product Details
MHCD0805
Species Reactivity
Published species
Host/Isotype
Class
Type
Clone
Immunogen
Conjugate
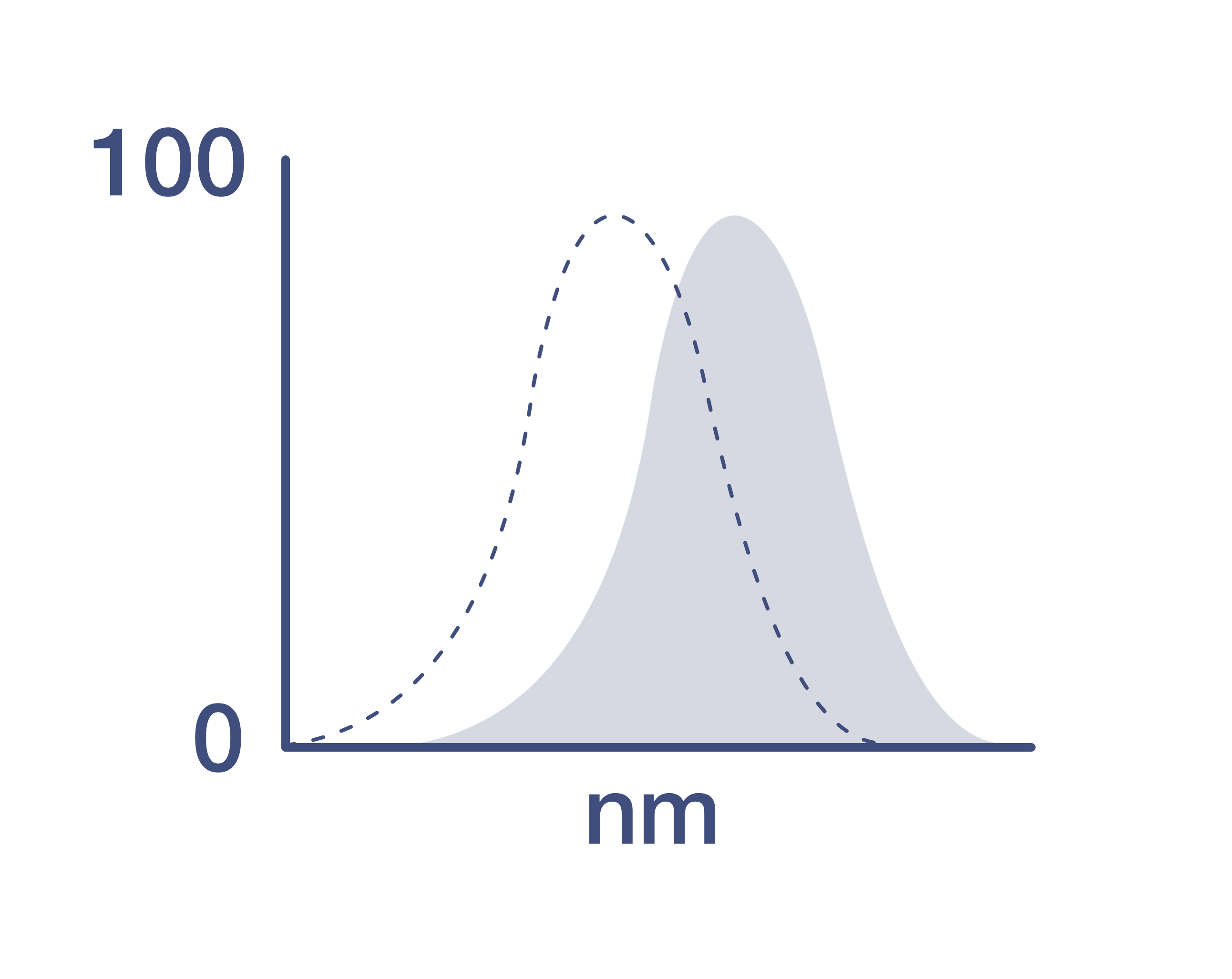
Excitation/Emission Max
Form
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Product Specific Information
Allophycocyanin (APC) is a stable and highly soluble phycobiliprotein that provides maximal absorbance and fluorescence without susceptibility to internal or external fluorescence quenching, thus providing exceptional quantum yields and molar extinction coefficients.
This antibody recognizes the alpha chain alone as well as the alpha/beta heterodimer.
Target Information
CD8, also known as cluster of differentiation 8, is a type I transmembrane glycoprotein of the immunoglobulin family that plays a crucial role in T cell differentiation, activation, and signal transduction. It is expressed as either a heterodimer (CD8 alpha beta) or a homodimer (CD8 alpha alpha). The CD8 alpha beta form is predominantly found on the majority of thymocytes and a subpopulation of mature alpha beta TCR T cells, while the CD8 alpha alpha form is expressed on gamma delta TCR T cells, a subset of intestinal intraepithelial lymphocytes (IELs), and dendritic cells. CD8 functions as a co-receptor for major histocompatibility complex class I (MHC-I) molecules, working alongside the T cell receptor (TCR). The CD8 alpha chain is essential for binding to MHC-I. CD8 is also expressed on a subset of T cells, NK cells, monocytes, and dendritic cells as disulfide-linked homodimers of CD8 alpha. Upon ligation of MHC-I/peptide complexes presented by antigen-presenting cells (APCs), CD8 recruits lymphocyte-specific protein tyrosine kinase (Lck), leading to lymphokine production, increased motility, and activation of cytotoxic T lymphocytes (CTLs). Activated CTLs are vital for clearing pathogens and tumor cells. The differentiation of naive CD8+ T cells into CTLs is strongly enhanced by cytokines such as IL-2, IL-12, and TGF-beta1. Through its interactions with MHC-I and association with protein tyrosine kinase p56lck, CD8 plays a significant role in T cell development and the activation of mature T cells.
Analyte Specific Reagent
How to use the Panel Builder
Watch the video to learn how to use the Invitrogen Flow Cytometry Panel Builder to build your next flow cytometry panel in 5 easy steps.
Bioinformatics
Protein Aliases: CD8 antigen, alpha polypeptide (p32); CD8 antigen, beta polypeptide 1 (p37); CD8a; CD8a molecule; CD8alpha; CD8b; CD8b molecule; CD8beta; cell surface glycoprotein T8 precursor; fCD8; Leu-2; leu-2a; Leu2 T-lymphocyte antigen; Ly-3 homolog; OKT8 T-cell antigen; T cell co-receptor; T lymphocyte surface glycoprotein (CD8-beta) precursor; T lymphocyte surface glycoprotein beta chain; T-cell antigen Leu2; T-cell differentiation antigen Leu-2/T8; T-cell surface glycoprotein CD8 alpha chain; T-cell surface glycoprotein CD8 beta chain; T-lymphocyte differentiation antigen T8/Leu-2; T8 T-cell antigen; unnamed protein product
Gene Aliases: CD8; CD8A; CD8alpha; CD8B; CD8B1; CD8beta; IMD116; LEU2; Ly-3; LY3; LYT3; MAL; p32; P37
UniProt ID: (Human) P01732, (Human) P10966
Entrez Gene ID: (Human) 925, (Human) 926

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support