With the rise of electromobility and the consequent increase in electric vehicle (EV) manufacturing, the market for lithium-ion batteries (LIB) has seen consistently high growth rates over the last decades. Heading toward zero emission goals in Europe and worldwide the global lithium-ion manufacturing capacity is expected to experience an unprecedented demand for battery raw material and manufacturing capacities in the upcoming decade. At the same time, battery research and quality control requirements will need to keep up with the accelerated demand for electric storage capacities.

In my recent blog post Challenges in Lithium-ion Battery Manufacturing and Quality Analysis – Part 1, I discussed the economic landscape in the lithium-ion battery market, growth forecast and analytical requirements in quality control and monitoring, as well as technologies involved in battery testing and material analysis. In this post I will take a deep dive into some applications and technologies that support quality control and testing of battery material.
Analytical requirements in quality control and monitoring
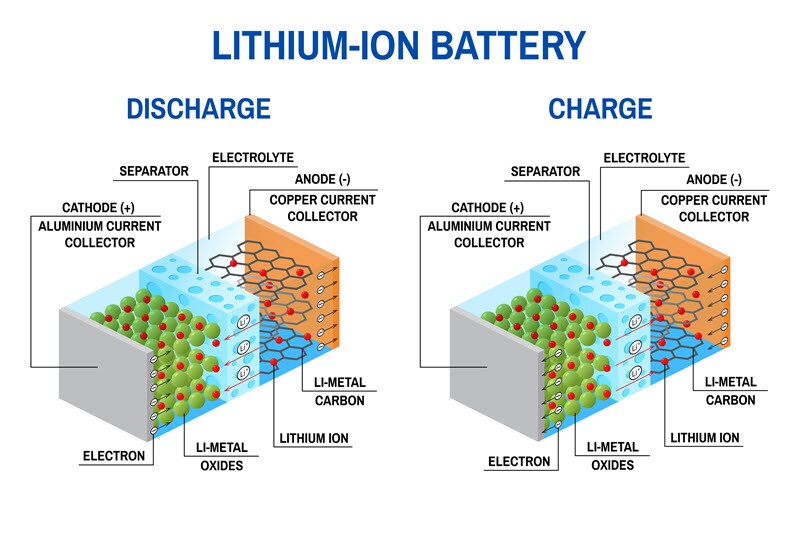
As mentioned in my previous blog post, there are three main components of a battery: two terminals made of different chemicals (typically metals), the anode and the cathode; and the electrolyte, which separates these terminals. The electrolyte is a chemical medium that allows the flow of electrical charge between the cathode and anode. The infographic below provides an overview of the battery structure, including Li-ions and electrons flow during charge and discharge. During manufacturing battery producers must not only deliver consistent overall quality, but they must also deliver it throughout the manufacturing process. Likewise, development of new battery materials must ascertain all the critical parameters that could affect battery performance throughout the entire manufacturing process. Let’s evaluate the main components of a battery, cathode, anode and electrolyte and investigate why quality is important to ensure performance.
Battery cathode material and elemental composition
A cathode’s active material is composed of lithium and, in the majority of the cases, one or several metals. Active materials have different characteristics depending on the type and proportion of metal in the cathode. For example, Ni (nickel) has high capacity, Mn (manganese) and Co (cobalt) have high safety, and Al (aluminum) increases the power of a battery. A cathode typically consists of a lithium transition metal oxide such as lithium-cobalt oxide (LiCoO2, LCO) and lithium-manganese oxide (LiMnO2, LMO) coated on metal foil (copper or aluminum).
The preferred cathode materials for EV batteries, on the other hand, are nickel-cobalt-aluminum oxide (Li(NiCoAl)O2, NCA), nickel-manganese-cobalt oxide (Li(NiMnCo)O2, NMC), and lithium-iron-phosphate (LiFePO4, LFP). It is fair to say, however, that battery development is a dynamic market and other types of batteries with a distinct cathode material structure are at the forefront, as for instance the Na-ion battery that was introduced by CATL in 2021.
Typically, mixtures of these materials are applied commercially with varying contents. Proportion and content of the main elements in cathode material can affect the performance and cost of the lithium battery significantly, and the content of impurities in the material alters the safety of the battery. Purity of the cathode material is critical, and changes in the raw material processing or synthesis can cause the introduction of impurities in the final cathode material. Conclusively, accurate determination and quantification of the main elements, as well as trace impurities, becomes particularly important.
Here the inductively coupled plasma optical emission spectrometry (ICP-OES) provides a rapid detection method for the determination of major elements and trace impurities in material used in lithium batteries. The application notes below demonstrate a fast analytical method for the determination of major and trace elements in the ternary cathode material of lithium batteries using the Thermo Scientific™ iCAP PRO Series ICP-OES. The notes describe the method development as well as presenting key figures of merit, such as detection limits and stability.
Application note: Sensitive determination of elements in lithium batteries using the ICP-OES
Application note: Determination of elemental impurities in lithium iron phosphate using ICP-OES
Battery anode material and elemental impurities
A state-of-the-art lithium-ion battery anode is commonly graphite-based. Though other carbon-based materials, such as graphene or silicon-based materials, tin-based materials, and metal oxides have been developed and may provide an alternative for next-generation batteries. Currently, most anode material is generally made from graphite powder.
Graphite powder is suitable for this application primarily because it is an easily molded, chemically stable, and non-metallic material with good electrical conductivity and high temperature, oxidation, and corrosion resistance. It also has a large lithium-ion diffusion coefficient with a high lithium insertion capacity and does not change volume with insertion of lithium ions. In addition, graphite powder can be modified through various oxidation and pyrolysis processes to generate a core-shell structure that can improve its charging/discharging performance and increase the anode lifetime. Therefore, graphite powder has become the main lithium-ion battery anode material in use today in smaller consumer goods, such as mobile phones, as well as in electric vehicles.
To ensure the quality of graphite as anode material, ICP-OES can provide a robust, accurate and reliable analytical method for the determination of various trace elements in graphite material for lithium-ion batteries. Application of ICP-OES provides the advantages of high sensitivity, good stability, fast analysis speed and low operating costs required for all aspects of elemental analysis in the lithium-ion battery workflow. The application note below demonstrates the performance of the Thermo Scientific™ iCAP™ PRO X ICP-OES Duo instrument for quantitative trace element impurity analysis in graphite powder samples used for lithium-ion battery anode production.

Electrolyte impurity analysis
Electrolyte plays a key role in transporting the positive lithium ions between the cathode and anode, and consequently the charging and discharging performance of the battery. Hence, it needs to be checked for potential impurities. At the same time the electrolyte is also a sample type that allows the investigation of ageing processes, as degradation products from all components of the battery can accumulate within it over time.
The electrolyte consists of a conducting salt in an organic solvent. The most common electrolyte salt is lithium hexafluorophosphate (LiPF6), but there are also lithium perchlorate (LiClO₄), lithium tetrafluoroborate (LiBF₄), lithium hexafluoroarsenate (LiAsF₆), lithium hexafluorosilicate (LiSiF₆), and lithium tetraphenylborate (LiB(C₆H₅)₄). The electrolyte in lithium-ion batteries is often a mixture of lithium salts and additional organic solvents. Some organic solvents used in the electrolyte solution are ethylene carbonate, diethyl carbonate, dimethyl carbonate, ethyl methyl carbonate, propylene carbonate, methyl formate, methyl acrylate, methyl butylate and ethyl acetate. Additives such as vinylene carbonate (VC) and fluoroethylene carbonate (FEC) are commonly added to the electrolyte to improve the overall performance of lithium-ion batteries.
Considering the chemical compound variety in the electrolyte (salts, ionic species, organic solvents, metals, etc.), different analytical techniques are required depending on analytical challenge. For instance, anions of lithium salts can be determined by ion chromatography (IC) to ensure that the solutions have been prepared at the proper concentrations. Gas chromatography or gas chromatography–mass spectrometry (GC-MS) can be used to provide the qualitative and quantitative composition of organic solvents in the electrolyte and aging byproducts. The inductively coupled plasma optical emission spectrometry (ICP-OES) provides robust and sensitive measurement of trace element impurities. Investigate the application notes below for a better understanding of how those techniques can be used for electrolyte analysis and quality testing.
Case study: Orbitrap GC-MS Technology Provides New Insight into Lithium Ion Battery Degradation
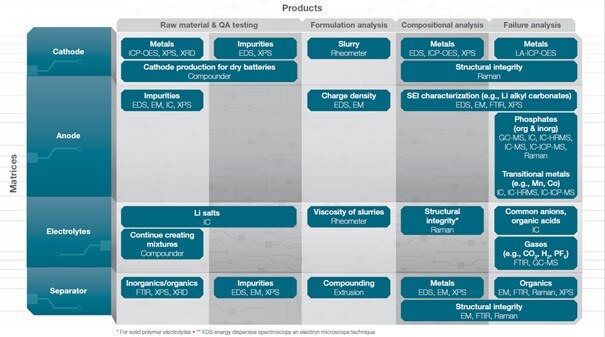
As you can see, many different analytical methods can be applied for quality testing of battery material. If you’d like a better overview on the available tools and techniques for chemical, elemental, and structural analysis, please visit our “Advanced battery technology enabled with Thermo Scientific tools and instruments” page or discover additional resources below.
Additional resources
Webinar: Powering Up Battery Materials Analysis using ICP-OES and ICP-MS
Website: Elemental Analysis Solutions for Battery Material Testing
Website: Battery technology research enhanced with electron microscopy and spectroscopy
Blog Post: Challenges in Lithium-ion Battery Manufacturing and Quality Analysis – Part 1
Blog Post: Is Battery Technology Just All About Lithium?
Blog Post: Making Energy Greener – What Role Does Elemental Analysis Play? Part 1



