Four examples investigating carbonaceous materials by Raman Spectroscopy for battery & tribological applications using a heating stage – and one word of warning of data integrity when using the Raman lasers
Turning unwanted items into materials for next-generation batteries, Dr. Vilas Pol’s ViPER group lab at the School of Chemical Engineering & School of Materials Engineering, Purdue University uses Raman extensively in the characterization of novel carbonaceous materials. In our recorded presentation, Professor Pol talks through four specific examples investigating carbonaceous materials by Raman spectroscopy for battery and tribological applications, including the use of an in-situ heated stage to monitor the properties of upcycled packing peanuts into battery anode materials.

Rechargeable Battery Diagnostics Using Raman Spectroscopy
Formerly at Argonne National Lab, Dr. Pol came to Purdue and started the ViPER Experimental Battery Lab. The work is focused on finding an alternative material to lithium for use in rechargeable batteries, and materials that the team is researching include sodium ion, potassium ion and lithium sulfide materials. Sometimes these materials turn out to be useful for many other applications, including tribological applications, as shown in Dr. Pol’s fourth example.
Batteries are a very complex system, and Dr. Pol shows us that Raman spectroscopy provides a fingerprint of the structural aspects of the molecules of battery material. “Raman spectroscopy provides great evidence if the material is pure or not.”
Dr. Pol’s lab uses a Thermo Scientific™ DXR Raman spectrometer for their experiments. “It is very user friendly; all the students love it,” Dr. Pol says at about eleven minutes into the webinar. “They get their data in a quicker fashion.” Their Raman spectrometer also comes with a temperature-controlled stage, and the team uses the stage to heat the sample and take Raman measurements during heating.
“Another beauty of technique,” Dr. Pol says about Raman, “is it requires small sample, and sample prep takes about a minute.” Additionally, the technique can be used in situ or ex situ, important for battery materials.
Dr. Pol describes four separate examples of using Raman Spectroscopy used to design and study the carbonaceous material created in his lab:
Example 1. Designing 3D Carbon Anodes
Pol’s first example is that of a starch-based precursor studied during in-situ heating. “The beauty of this process is you can tune the porosity of the material itself based on how fast you are heating it up.” Micron-sized pored of 10-15 um can be achieved via heating rate, and if heated slowly, most of the pores stay closed.
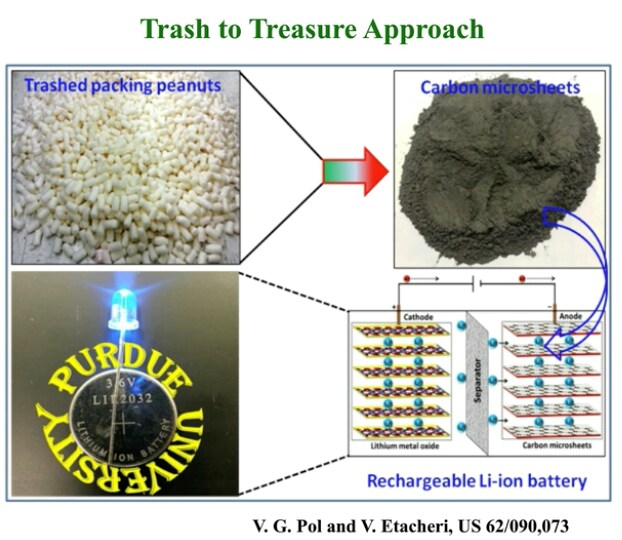
Example 2 – Upcycling Packing Peanuts
In Pol’s second example, he describes how battery material can be made of packing peanuts. “We made carbonaceous material out of both” (expanded polystyrene based and starch based), “but I will only give examples of the starch based that is similar to the previous studies where we utilized starch and here we get a unique architecture that is sheet-like carbon that is a very good electron material for the sodium ion battery application.”
Rechargeable Battery Diagnostics Using Raman Spectroscopy
The material is heat treated to keep a specific mixture of sp2 and sp3 carbon bonds, which play a key role in the resulting microstructure for the sodium ion to shuttle back and forth within the matrix. Once the material is heat-treated, a highly porous architecture of carbon is achieved. “It is a high-purity carbon,” as shown by the Raman, Pol says, “And the material is ready to use.”
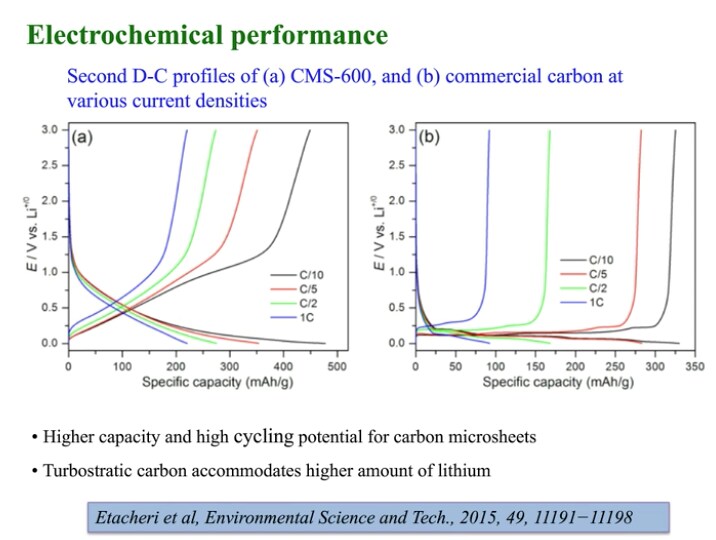
Using both Raman and scanning electron microscopy, the team found that the material is like a three-dimensional matrix of carbon microsheets (CMS). By adjusting the temperature during heat-treatment, the electrochemical performance of the material can be tuned. (Shown in the “electrochemical performance” slide shown here.) The performance also shows that, “this can make the batteries safer.”
Rechargeable Battery Diagnostics Using Raman Spectroscopy
Example 3 – Novel Carbon Spheres for Li-ion Batteries
The effect of pyrolysis temperature on the carbon morphology of these materials, and the resulting battery performance, was studied via Raman spectroscopy in Pol’s third example. For this experiment, the material was heated ex-situ to extremely high temperatures – 600, 900, 1500, and 1800 degrees Celsius. As the material is heat treated, the surface area of the material goes down.

Rechargeable Battery Diagnostics Using Raman Spectroscopy
Example 4 – Carbon materials for Tribology Applications
In his fourth example, Dr. Pol details the effects of carbon spheres with molybdenum sulfide additives for use in tribology applications. The molybdenum sulfide nanocoating results in excellent tribological performance.

Rechargeable Battery Diagnostics Using Raman Spectroscopy
Further information about the work being done in the ViPER (Vilas Pol’s Energy Research) lab can be found here.
Postscript –

While not part of Dr. Pol’s webinar, it might be interesting for the reader to learn that this past August, Prof. Vilas Pol set a new Guinness World Record for the fastest time (8 minutes 36 seconds) to arrange all elements of the periodic table. A link to this accomplishment can be found via the ViPER lab website. Congratulations, Prof. Pol from your friends at Thermo Fischer Scientific!




Leave a Reply