Search
Practical Tips for Optimal Northern Analysis
Northern assays require RNA (total- or poly(A)- selected) to be resolved on a agarose gel under denaturing conditions, transferred to a membrane and immobilized for subsequent hybridization. Various inconsistencies in the procedure, particularly the transfer set-up, can greatly effect the assay's outcome. Here, we describe several technical points which can make any Northern analysis a success.
Gel Parameters - Less is More
- Pour gels as thin as possible. The gel must be thick enough to provide well depth to accommodate sample volume, but the thinner the gel, the faster and more efficient the transfer will progress.
- Transfer is also impeded by agarose gel concentrations greater than 1.2%.
Gel Transfer to Membrane
Transfer of RNA to a membrane (Northern blotting) can be accomplished using capillary transfer or electroblotting. Electroblotting utilizes a manufactured apparatus and is extremely efficient as long as the manufacturer's instructions are followed. Capillary transfer utilizes a stack of dry paper towels to draw the transfer solution though a wick of blotting paper (such as Whatman 3MM), the gel, the membrane and into the dry towels. The capillary transfer set up can be upward or downward (Figure 1). Regardless of the method chosen there are hints that can optimize any capillary transfer.
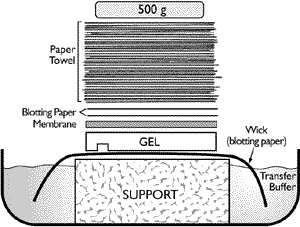
- Measure twice, cut once—Capillary transfer requires not only a membrane cut to the size of the gel, but also several sheets of blotting paper and a stack of paper towels several centimeters in height. To conserve transfer solution and materials, it is useful to cut any excess gel away from your samples and measure the gel prior to cutting blot materials. A standard office paper cutter aids this process considerably. The stack of paper towels, which should be three or more centimeters high, is easily cut using a sharp razor blade or utility knife and a straight edge.
- Hands off—When cutting the membrane, it is useful to keep the protective paper on either side of the membrane while cutting and handling. Otherwise, the membrane should be handled only by its edges and with forceps. Fingerprints or gloveprints can contribute to background signal seen on autorads, this is especially true for membranes subjected to nonisotopic detection.
- Get oriented—It is useful to mark the membrane for orientation prior to wetting; e.g. cut off a corner or write the date on the upper right corner. The date can be lightly marked with a pencil on the back of the membrane (opposite side from the RNA).
- Tiny bubbles—When setting up the transfer take care not to introduce bubbles in any of the layer; it is especially important to avoid bubbles between the gel and membrane. As each component is added to the blot, a pipette can be used to gently roll out any bubbles.
- Dodging the issue—It is important that the transfer not be "short-circuited". This occurs when any of the blotting materials come into contact with the transfer solution wick. For efficient transfer, the solution must only pass through the gel and membrane with no route around the two. To avoid "short circuiting", cut all materials to the size of the gel and mask around the gel with strips of parafilm or plastic wrap.
- Bottoms up (or down, as the case may be)—Be sure the side of the gel corresponding to the bottom of the wells is against the membrane. This minimizes the distance the RNA must migrate out of gel to the membrane.
Cross Linking
After transfer is completed the membrane should be treated to immobilize the nucleic acid, otherwise there is a loss in signal intensity (Figure 2). There are several ways in which this is accomplished, including baking in a vacuum or conventional oven (30 min to 2 hours at 80°C), UV crosslinking or microwaving. At Ambion, we find UV crosslinking to be the most convenient and effective method.

Figure 2. Effect of various methods of crosslinking on Northern sensitivity. Replicate dilutions of rat thymus total RNA were electrophoresed and transferred to BrightStar-Plus Positively Charged Membrane according to the NorthernMax protocol. The membrane was cut into three pieces and the RNA on each blot was crosslinked by the method described. The blots were hybridized to an RNA probe complementary to rat Beta-actin at a concentration of 1 x 106 cpm probe/ml NorthernMax Hybridization Solution at 68°C overnight, washed and exposed to the same piece of film.
Should RNA Samples in Northern Analysis Be Stained With Ethidium Bromide?
Whether or not a Northern gel should be stained with ethidium bromide (EtBr) is a point of debate. Most sources recommend staining with EtBr after electrophoresis to check sample integrity followed by destaining prior to transfer. Staining after the gel has been run is recommended because EtBr in the samples can supposedly alter migration of the RNA and inhibit complete transfer of the RNA to the membrane, resulting in a loss in sensitivity. However, it is extremely convenient to add EtBr to your samples prior to electrophoresis. This allows for several additional checks in the procedure. The migration distance and integrity of the RNAs can be monitored during electrophoresis and the degree of transfer can be easily assessed by viewing the membrane and gel on a UV transilluminator (Figure 3). Furthermore, the position of markers, and ribosomal RNA bands can be marked on the membrane post-transfer with a pencil. Equivalent EtBr staining across samples can serve as an internal control for RNA sample loading. The presence of EtBr does slightly alter mobility of the RNA through the gel, as seen in Figure 4. Therefore, if EtBr is added to the samples prior to gel electrophoresis, it should be added to all samples; adding EtBr to a single lane will provide information about RNA integrity but should not be used as a sizing marker.
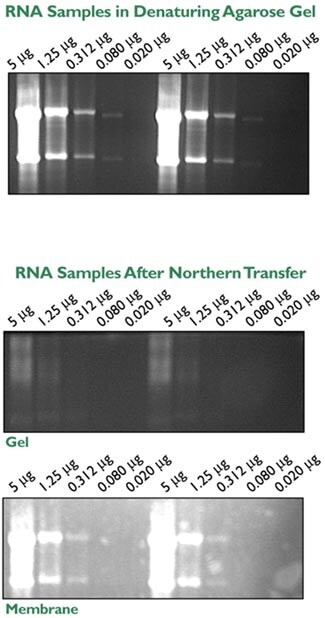
Figure 3. Use of Ethidium Bromide Staining to Monitor Electrophoresis and Northern Transfer. Dilutions of rat thymus total RNA were mixed with NorthernMax Gel Loading Solution containing 10 µg/ml EtBr and electrophoresed according to the NorthernMax protocol. The gel was photographed under UV light at an exposure of 12/30 seconds (Top Panel). The samples were transferred to BrightStar-Plus Positively Charged Nylon membrane using the NorthernMax 1-hour capillary transfer protocol. Post transfer the gel and membrane were photographed under UV light at an exposure of 20/30 seconds (Middle and Bottom Panels).
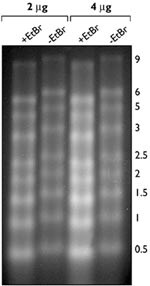
Figure 4. Effect of Ethidium Bromide on RNA Migration in a Denaturing Agarose Gel. 2 or 4 µg of RNA Millennium Markers™ were mixed with NorthernMax Gel Loading Solution with or without 10 µg/ml EtBr. Samples were heated and electrophoresed according to protocol. After electrophoresis, the gel was stained with EtBr for one hour, destained with water and photographed under UV light. Note, RNA bands diffused due to the lengthy staining and destaining process.
At Ambion, we have seen a slight decrease in signal intensity with formaldehyde gels of samples containing EtBr compared to samples with no EtBr (Figure 5). To minimize this decrease we recommend using 10 µg EtBr/mL gel loading solution.
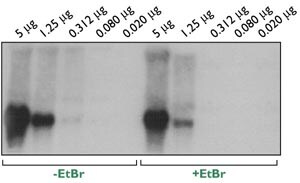
Figure 5. Effect of Ethidium Bromide on Northern Sensitivity. Replicate dilutions of mouse spleen total RNA were prepared; one set with 10 µg/mL EtBr added to NorthernMax Gel Loading Solution and the other set without EtBr. The samples were run and blotted according to Ambion's NorthernMax procedure. An RNA probe complementary to cyclophilin message was generated with the Strip-EZ RNA Probe Synthesis Kit. Hybridizations were done with 1 x 106 cpm probe/mL NorthernMax Hybridization Solution at 68°C overnight. The film was exposed at -80°C for 5 days with one intensifying screen.
Overall, the benefits of including EtBr in Northern samples probably outweigh the minor loss of signal intensity and effects on RNA migration. Adding EtBr to samples should be avoided only if required by an extremely rare message.
For Research Use Only. Not for use in diagnostic procedures.