What happens when a multiplex qPCR assay begins to lose performance as additional targets are introduced? An assay that performs reproducibly in singleplex can exhibit Ct shift, reduced sensitivity, or compressed dynamic range when scaled to 5- or 6-plex.
As plex level increases, competition among primers, probes, reporter dyes, and polymerase intensifies, and small imbalances can become amplified under multiplex conditions. Modest ΔCt changes may affect quantification accuracy if reaction components are not designed to function together under multiplex conditions. Successfully scaling multiplex qPCR requires more than combining additional assays within the same reaction; it requires coordinated optimization of probe chemistry, dye compatibility, and master mix formulation.
What you’ll learn in this article
To scale confidently from 1-plex to 6-plex without compromising sensitivity, it is important to understand where multiplex workflows typically lose performance and how to mitigate those effects.
In this article, we’ll cover:
- Why ΔCt shift occurs during multiplex scale-up
- How probe chemistry influences signal strength and spectral separation
- The role of master mix formulation in maintaining amplification efficiency and dynamic range
- Considerations for preserving low-copy detection in higher-plex reactions
- Practical steps to evaluate and optimize 5- and 6-plex assays
Explore additional information including the chemistry and workflow considerations that support reliable high-order multiplex qPCR development.
What is multiplex qPCR?
Multiplex qPCR is defined as the amplification of more than one target in a single reaction, using different reporter dyes to distinguish each target.
In typical workflows:
- Each assay/target pair is labeled with a distinct reporter dye
- All assays are combined with a single master mix and template
- Amplification is detected across separate optical channels
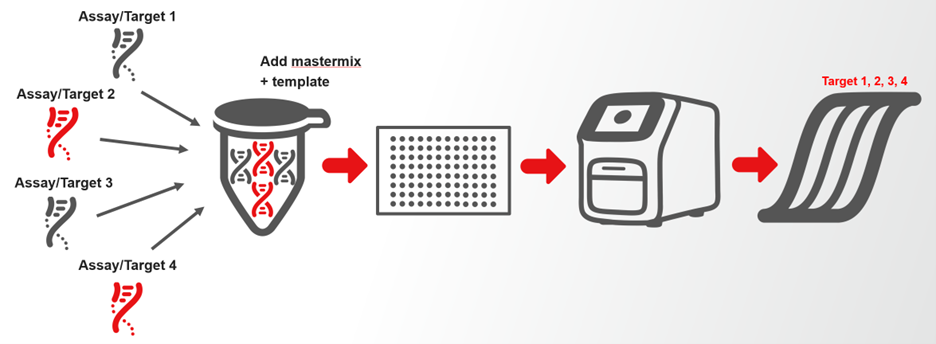
Multiplexing can also involve the amplification of multiple targets with a single channel, depending on assay design.
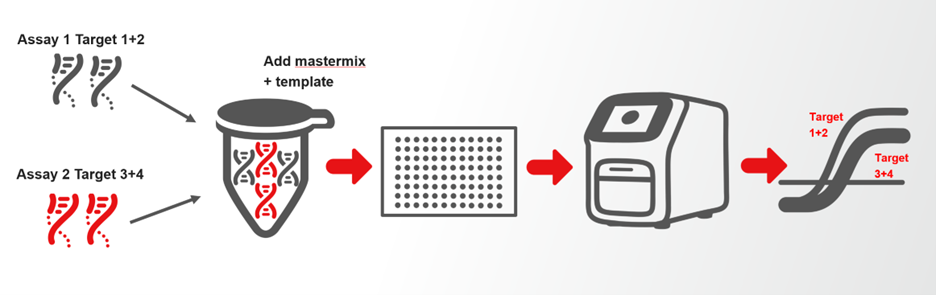
When properly designed, multiplex qPCR may enable:
- Reduced reagent and sample usage
- Increased throughput
- Shorter workflow time
- Cost-effective for multiple targets
- Improved precision when comparing targets within the same reaction
However, these advantages depend heavily on assay design and reaction optimization. Understanding what multiplex qPCR is only the starting point. Performance at higher plex levels depends on how the assay is designed and how reaction components interact.
General considerations for multiplex assay design
Successful multiplex qPCR workflows require alignment across several variables:
- Application and sample type
- Primer and probe design
- Master mix compatibility
- Thermal cycling conditions
- Instrument and dye compatibility
Specific assay design considerations include:
- Minimizing nonspecific amplification
- Selecting dye/quencher combinations that reduce background and cross-talk
- Optimizing annealing temperature and extension time across all targets
- Limiting competition or inhibition between assays
- Accounting for expression range differences among targets
- Optimizing primer and probe concentrations to balance amplification efficiencies and reduce polymerase saturation
These parameters become increasingly critical as plex level rises.
How probe chemistry influences multiplex performance
Probe chemistry directly affects signal clarity, background suppression, dynamic range, and scale-up performance. Applied Biosystems™ QSY™ probe portfolio with TaqMan™ QSY2™ probes, supports higher-order multiplexing.
Key characteristics include:
- Higher analytical sensitivity
- Wider dynamic range
- Improved performance retention when scaling from 1-plex to 6-plex
- Smaller ΔCt difference between 1-plex and 6-plex reactions compared to certain alternative probes
QSY2 probes support high-throughput 5- and 6-plex reactions using Cyanine 5 and Cyanine 5.5 reporter dyes, helping expand multiplex panel capacity while maintaining signal separation. Probes must be redesigned when switching between MGB and QSY quenchers, reinforcing that dye-quencher pairing is not interchangeable.
For custom multiplex panel development, Applied Biosystems™ Custom Primers & Probes provide configurable dye and quencher options support up to six targets in a single reaction.
Why ΔCt retention matters in 6-plex reactions
A key performance metric is ΔCt retention when scaling from singleplex to multiplex. TaqMan QSY2 probes have demonstrated smaller ΔCt differences between 1-plex and 6-plex reactions in internal performance evaluations compared to selected alternative probe chemistries.
Maintaining a small ΔCt during scale-up helps preserve:
- Quantification confidence
- Low-copy sensitivity
- Dynamic range stability
ΔCt drift during scale-up often reflects reaction imbalance rather than primer design alone.
The role of master mix formulation in multiplex stability
Assay design alone does not determine multiplex success. Master mix formulation is a primary contributor to performance retention under multiplex load. The Applied Biosystems™ TaqPath™ portfolio includes master mixes verified for multiplexing applications including TaqPath™ DuraPlex™ 1-Step RT-qPCR Master Mix with Rox and no Rox designed and optimized for higher-order multiplexing.
Preserving performance in complex sample matrices
Multiplex qPCR performance must be maintained across diverse and sometimes inhibitory sample types, including blood-derived matrices, viral transport media, and biologically complex specimens.
Key workflow considerations include:
- High tolerance to inhibitors commonly found in clinical research or biological samples
- Stable amplification across varying template inputs
- Compatibility with automated liquid handling systems
- Benchtop stability of assembled reactions for high-throughput workflows
- Consistent lot-to-lot performance to support longitudinal studies
What is the best way to optimize multiplex qPCR for 6 targets?
After understanding the chemistry and reaction dynamics, optimization should prioritize analytical reproducibility, low-copy sensitivity, and robustness across sample types. Effective multiplex qPCR optimization generally includes:
- Establishing baseline singleplex assay efficiency
- Comparing ΔCt between singleplex and multiplex formats
- Confirming slope consistency and dynamic range retention
- Evaluating analytical performance across relevant input ranges
- Assessing inhibitor tolerance using representative sample matrices
- Confirming reproducibility across reagent lots
Successful 6-plex performance depends on two major factors:
- Assay design (primer/probe chemistry, dye-quencher pairing)
- Master mix formulation optimized for multiplexing
Probe chemistries such as TaqMan QSY and QSY2 Probes are engineered to support higher-order multiplexing with improved performance retention during scale-up, while master mixes such as TaqPath DuraPlex are formulated to support simultaneous multi-target amplification with broad dynamic range.
This combination can help maintain analytical confidence when expanding panels for gene expression profiling, viral detection, or biomarker evaluation.
Ready to optimize multiplex qPCR?
For a practical, step-by-step approach to optimizing multiplex reactions, including guidance on assay balancing and scale-up evaluation, read the TaqMan™ Multiplex PCR Optimization Guide.
Download the TaqMan Multiplex PCR Optimization Guide to support assay development and scale-up.
For additional data and assay design insights discussed in this article, watch the Mastering Multiplexing webinar
Explore qPCR multiplex-ready assays and master mixes designed for high-order multiplexing:
Browse TaqMan Multiplex probes and qPCR primers
Shop TaqMan MGB Probe for 5-plex multiplexing
Design a custom multiplex assay
Shop TaqPath DuraPlex 1-Step RT-qPCR Master Mix
Speak to a multiplexing specialist
Build a reliable 6-plex workflow with probes and master mixes designed for high-order qPCR multiplex performance.
TaqMan Predesigned Gene Expression Assays
Custom TaqMan Assays
TaqMan QSY and QSY2 Probes
TaqPath DuraPlex 1-Step RT-qPCR Master Mix
QuantStudio Real-Time PCR Systems
For Research Use Only. Not for use in diagnostic procedures.



Leave a Reply