As you gather around with friends and family this holiday season, we at Thermo Fisher Scientific hope you will give a moment to reflect on one of most useful techniques in material science and quality control: spectroscopy. Besides lab instruments or portable analyzers, spectrometers can be found in many different places. Want to know how much sugar is in a single M&M candy? Use a spectrometer with diffuse reflection. Want to know what elements a star emits? Use a telescopic spectrometer. Want to label all the colors in Mom’s ugly sweater? Use your eyes! That’s right, your own eyesight is a spectrometer.
Rudolph the Red-Nosed Reindeer
What is infrared radiation?
On Tuesday, December 17, 2019 at 11 am EST (4 pm GMT) we present an informative, but informal discussion asking a few questions about infrared spectroscopy, like if Rudolf’s red nose shines bright infrared radiation, what would Rudolf see? Infrared radiation sits in the middle of the electromagnetic spectrum. But what is infrared radiation and what exactly is the electromagnetic continuum? We shall explain.
It’s all about photons
Electromagnetic radiation can be described in terms of a stream of particles with no electrical charge, called photons. They travel in a wave-like pattern at the speed of light. Each photon contains a certain amount of energy. The different types of radiation are defined by the amount of energy found in the photons, starting with radio waves with low energies. Microwave photons have a little more energy, infrared photons have still more, then visible, ultraviolet, X-rays, and, the most energetic of all, gamma-rays.
Photons are elementary particles, meaning that they are the smallest piece of light possible. In physics, energy is the property that is transferred to an object in order to perform work, such as creating heat. Common forms of energy include the kinetic energy of a moving object, the potential energy stored by an object’s position in gravitational, electric or magnetic forces, the elastic energy stored by stretching solid objects, the chemical energy released when a fuel burns, the radiant energy carried by light, and the thermal energy of an object’s temperature.
A photon’s energy is equal to its frequency multiplied by an important number— the Planck constant. This number is the minimum amount of an electromagnetic action, known as quantum. Or put another way: the minimal element of the energy of the electromagnetic wave itself, which is energy multiplied by time. This number describes the behavior of particles and waves on the atomic scale, including the particle aspect of light. It is the fundamental number is quantum mechanics and has led to our understanding of atomic structures and the energies of each element that is so useful in spectroscopies.
Electricity and magnetism
Magnetism and electricity are different aspects of the same force. Photons have energy proportional to their frequency and two possible polarization states. The electric field surrounding its charge oscillates, generating an oscillating magnetic field. The result is an electromagnetic wave. The speed with which an electromagnetic wave travels is the speed of light.
Wavelength quantifies the periods of oscillating currents of the electric and magnetic fields of the radiation (i.e., waves), and measured by frequency, which are the number of complete wave sets passing through a point at a certain time—typically measured as waves per second, or hertz. Whereas frequency can be thought of as the number of waves per unit time, wavenumber is the number of waves per unit distance, typically inverse centimeters (cm-1). Inverse cm is a wonderful way to measure light–the higher the wavelength, the greater the energy.
Frequency and wavelength are inversely related. Electromagnetic waves with higher frequencies, X-rays and gamma-rays, have shorter wavelengths. Consequently, electromagnetic waves like radio waves and microwaves have lower frequencies and longer wavelengths. Each portion of the electromagnetic spectrum has its special purposes. For humans, the most purposeful portion of this spectrum is light.
Electromagnetic radiation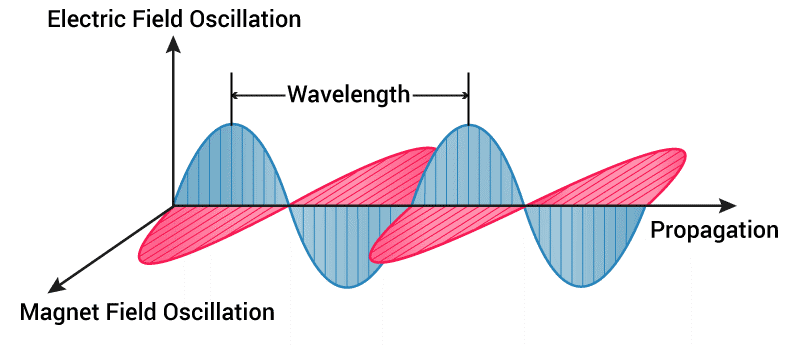
Visible light as electromagnetic radiation
Usually when we use the term light we generally mean visible light, but it can also represent ultraviolet and infrared light–whose names actually describe their positions on the electromagnetic spectrum—a number greater than the color violet (ultraviolet) and less than the color red (infrared). Most people can perceive light at roughly 400-700 nm in wavelength (25,000-14,286 cm-1 wavenumber). The infrared range covers 700-1000 nm (wavelength), or 14,286-12,800 cm-1 (wavenumber), and ultraviolet radiation has wavenumbers above these, approximately, 25,000 – 50,000 cm-1, or 100 to 400 nm (wavelength).
An infrared spectrometer detects photons that are transmitted or absorbed when a sample is excited with infrared light. Understanding the spectral pattern of peaks at various wavenumbers allows us to determine the molecular composition of the sample. Somewhat similar to an infrared spectrometer, our eyes are instruments that sort the wavelengths of visible light in order to identify color.
Our eyes are spectrometers
Human eyesight consists of photoreceptors, which are specialized cells that convert light into electrical signals. When photons reach receptor cells that contain a protein that releases a biochemical energy when excited at a certain wavelength. Rod cells are extremely sensitive to light and are useful for monochromatic vision in the dark. Three cone cells manage color vision. Each cone sends signals to the brain to calculate colors based on the individual photoreceptor and the intensity (count) of photons receives. Shapes are determined by the location of the cell on the retina while lenses determine distance and the iris regulates light intensity—much like chemical imaging in microspectroscopy.
Light radiation is both beneficial and potentially harmful
Radiation from the sun covers all of the electromagnetic spectrum, but mostly infrared, visible and ultraviolet light. Because this radiation is the transfer of energy, it creates heat. Owing to the distance of our planet to the sun and filtering mechanisms in the atmosphere, this radiated heat arrives at an intensity that enables habitation. It also creates photochemical reactions that are essential to life. We know, too, that some radiation can have detrimental effects. While productive to the maintenance of the ozone layer and the synthesis of vitamin D, ultraviolet light directly damages DNA and causes sunburn.
Infrared light and climate
Infrared light is one of the most useful forms of electromagnetic radiation. Where there is insufficient visible light, infrared enables night vision and low light devices for cameras. Infrared signals are used in certain types of data communication including television remote controls. IR radiation is used for heating in both industrial and consumer processes. In addition to night vision goggles, IR radiation has several uses in military devices for missile guidance, homing and heat seeking.
Most of the earth’s heat comes from the absorption of infrared radiation. (Visible light is not absorbed or heated by the atmosphere.) Greenhouse gases, including water vapor, carbon dioxide, and methane, absorb and emit radiant energy within the thermal infrared range to maintain the Earth’s temperature. The balance between absorbed and emitted infrared radiation can affect the overall global temperature and climate.
Putting infrared radiation to good use
The range of infrared region is 12800 ~ 10 cm-1 and can be divided into near-infrared region (12800 ~ 4000 cm-1), mid-infrared region (4000 ~ 200 cm-1) and far-infrared region (50 ~ 1000 cm-1). In infrared spectroscopy, samples exposed to infrared radiation selectively absorb radiation of specific wavelengths, causing a change of dipole moment (separation of positive and negative charges) of sample molecules. Consequently, the vibrational energy levels of sample molecules transfer from ground state to excited state. The frequency of the absorption peak is determined by the vibrational energy gap. The number of absorption peaks is related to the number of vibrational modes of the molecule. The intensity of absorption peaks is related to the change of dipole moment and the possibility of the transition of energy levels. Therefore, by analyzing the infrared spectrum, one can readily obtain abundant structure information of a molecule.
The commonly used region for infrared spectroscopy is 4000 ~ 400 cm-1 because the absorption radiation of most organic compounds and inorganic ions is within this region. The region between 400 cm-1 and 1500 cm-1 is known as the fingerprint region, so called because it’s difficult to assign all the absorption bands, and because of the unique patterns found there. Absorption bands in the 4000 to 1450 cm-1 region are usually due to stretching vibrations of diatomic units, and this is sometimes called the group frequency region.
Webinar Presentation on FTIR
Just in time for the 2019 holiday season, we conduct an informative webinar presented by Dr. Mike Bradley. We will answer several burning questions such as:
- What are the various sampling techniques for Fourier-Transform infrared spectroscopy (FTIR)?
- How does FTIR verify compounds?
- What would an infrared-nosed reindeer be able to see?
Register for a live broadcast on Tuesday, December 17, 2019 at 11 am EST (4 pm GMT)
Want to speak to someone? Need a quote? Want a demo? |
Opt in for email communications on this and other topics. |
cool