Our previous metals article described how XPS helps manufacturers maintain the quality of high performance materials. X-ray Photoelectron Spectroscopy (XPS) is a fundamental characterization tool for investigating a wide range of surface problems on metal and oxide surfaces, and the ideal tool for measuring composition and thickness of protective oxide films on metals.
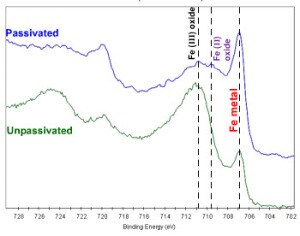
A recent webinar demonstrates how to identify passivation issues using XPS. The iron chemistry of passivated and unpassivated steels was investigated using high energy resolution XPS. Metallic and oxide states are observed in both cases, with significantly reduced oxidation detected on the passivated sample. However, the chromium oxide thickness is also reduced on the passivated sample. This reduction in total oxide thickness means that more of the iron-rich metallic substrate is seen by the surface XPS measurement – the lower-than-required values of total Cr to Fe and Cr-O to Fe-O reflect this effect.
Visual inspection of the XPS data reveals that passivation had significantly reduced the level of iron oxidation on the steel surface. Quantitative measurement of a number of useful parameters, however, demonstrated that the passivation method had not produced a protective layer of the required quality. Sputter profiling of the surface revealed that the passivating chromium oxide layer was too thin.
To see complete test data, including the correlation curves, repeatability data, methodology, and comments, view the webinar recording: XPS Simplified: Understanding Metal Surfaces and Oxides with X-ray Photoelectron Spectroscopy (XPS).






Leave a Reply