 This blog often discusses the advantages of utilizing portable X-ray Fluorescence (XRF) technology for providing immediate, actionable data for mining and exploration. But what is XRF, and how does it work?
This blog often discusses the advantages of utilizing portable X-ray Fluorescence (XRF) technology for providing immediate, actionable data for mining and exploration. But what is XRF, and how does it work?
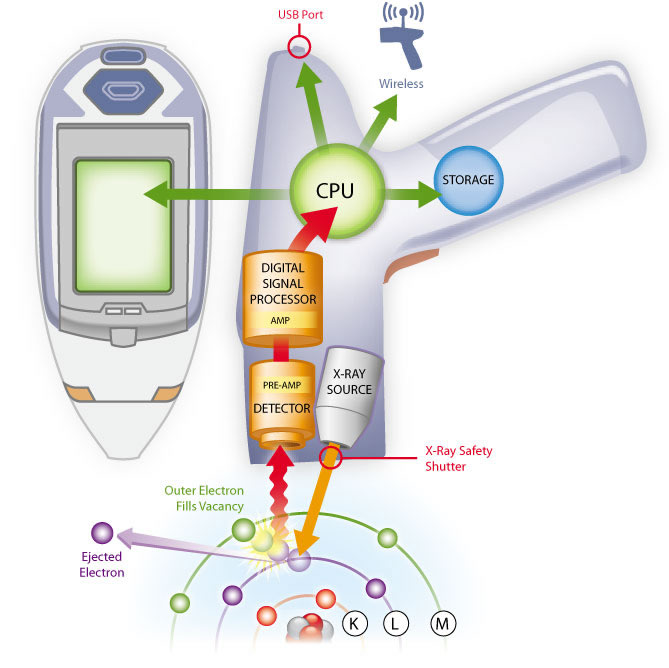
XRF is a non-destructive analytical technique used to determine the elemental composition of materials. XRF analyzers determine the chemistry of a sample by measuring the fluorescent (or secondary) X-ray emitted from a sample when it is excited by a primary X-ray source. Because this fluorescence is unique to the elemental composition of the sample, XRF is an excellent technology for qualitative and quantitative analysis of the material composition. To the right is an illustration of how a handheld XRF instrument works.
The X-ray Fluorescence Process
- A solid or a liquid sample is irradiated with high energy X-rays from a controlled X-ray tube.
- When an atom in the sample is struck with an X-ray of sufficient energy (greater than the atom’s K or L shell binding energy), an electron from one of the atom’s inner orbital shells is dislodged.
- The atom regains stability, filling the vacancy left in the inner orbital shell with an electron from one of the atom’s higher energy orbital shells.
- The electron drops to the lower energy state by releasing a fluorescent X-ray. The energy of this X-ray is equal to the specific difference in energy between two quantum states of the electron. The measurement of this energy is the basis of XRF analysis.
Interpretation of XRF Spectra
Each of the elements present in a sample produces a set of characteristic fluorescent X-rays that is unique for that specific element, which is why XRF spectroscopy is especially useful for elemental analysis. This elemental “fingerprint” is best illustrated by examining the X-ray energy spectrum and its “scattering peaks.” Most atoms have several electron orbitals (K shell, L shell, M shell, for example). When X-ray energy causes electrons to transfer in and out of these shell levels, XRF peaks with varying intensities are created and will be present in the spectrum. The peak energy identifies the element, and the peak height / intensity is generally indicative of its concentration.
Considerations
Phenomena that can influence the results of XRF analysis include scattering, which occurs when incoming X-rays do not produce fluorescence, but rather “collide” with the atoms of the sample which results in a change in the direction of motion of a particle. Spectral effects occur when elements have lines that overlap other elements. Matrix effects include enhancement effects, which occur when some fluorescent X-rays have more energy than the binding energy of other elements present in the sample, and absorption effects, which occur when a fluorescent X-ray does not reach the detector as it is scattered or absorbed by other elements present in the sample.
XRF Applications in Mining
Handheld XRF analyzers are a reliable method to analyze ore samples in open pits and underground mines – achieving the accuracy required to provide defensible information for process oversight, quality assurance, and various other operational decisions (such as grade control). Portable XRF technology can help ascertain the viability of lower grade resources and find localized high-grade enrichments, delineate ore from waste boundaries to reduce the randomness of digging, and obtain defensible data and minimize the need to send samples to external testing labs.
Sample analysis with handheld XRF analyzers offers a substantial advantage in mining operations by providing immediate feedback for quick decision making on site:
- Whether to stop or continue drilling
- When to make equipment relocation decisions
- Where to focus on the grid
- When to select a sample for laboratory analysis.
Real-time analysis with handheld XRF analyzers is also a good way to prequalify samples for off-site lab analysis to ensure only the best samples are evaluated. Portable handheld analyzers can be operated virtually anywhere on site and easily accommodate a wide variation of samples, with little or no sample preparation. XRF analyzers also quickly deliver exploration data for quantitative geochemical analysis of metal concentrations for geochemical mapping. If you want a quick read, but more information, about Handheld XRF and how it works, download our free ebook: Portable XRF Technology for the Non-Scientist.
Additional Resources
- ebook: Portable XRF Technology for the Non-Scientist
- Online resources for Mining & Minerals
Editor’s Note: This article was originally authored by Esa Nummi and published in April 2015, but has been updated and refreshed.







Leave a Reply