Search
Thermo Scientific Chemicals
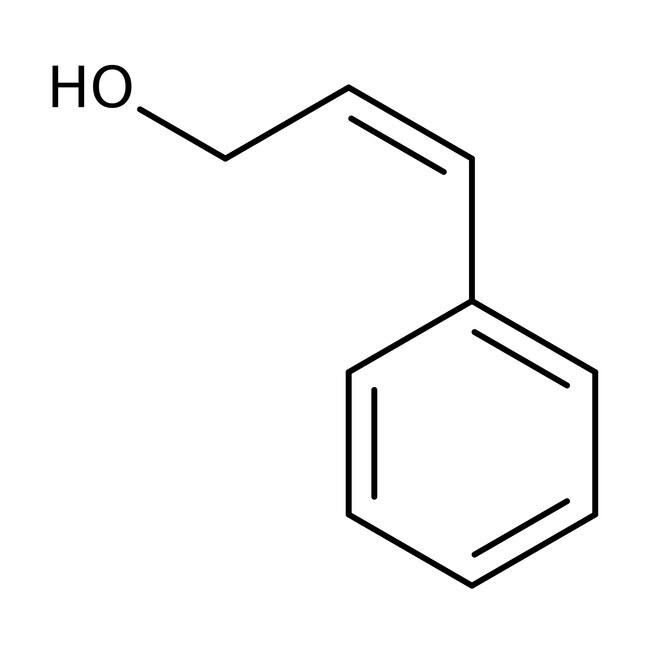
Cinnamyl alcohol, 98%
CAS: 104-54-1 | C9H10O | 134.178 g/mol
Catalog number A13025.30
also known as A13025-30
Price (JPY)Request A Quote
-
Quantity:
250 g
Chemical Identifiers
CAS104-54-1
IUPAC Name(2Z)-3-phenylprop-2-en-1-ol
Molecular FormulaC9H10O
InChI KeyOOCCDEMITAIZTP-DAXSKMNVSA-N
SMILESOCC=C/C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream to pale yellow
FormFused solid
Identification (FTIR)Conforms
Melting Point (clear melt)30.0-37.0?C
Assay (GC)≥97.5%
Cinnamyl alcohol is an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. It is also used in perfumery and as well as a deodorant.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Cinnamyl alcohol is an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. It is also used in perfumery and as well as a deodorant.
Solubility
Soluble in water 1.8 g/L (20°C).
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Stable under recommended storage conditions. Incompatible with strong oxidizing agents.
Cinnamyl alcohol is an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. It is also used in perfumery and as well as a deodorant.
Solubility
Soluble in water 1.8 g/L (20°C).
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Stable under recommended storage conditions. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- E Armengol.; ML Cano.; A Corma.; H García. Mesoporous aluminosilicate MCM-41 as a convenient acid catalyst for Friedel-Crafts alkylation of a bulky aromatic compound with cinnamyl alcohol. Journal of the Chemical Society, Chemical Communications.19957 (1), 519-520.
- RL Mansell.; GG Gross.; J Stöckigt.; H Franke.; MH Zenk. Purification and properties of cinnamyl alcohol dehydrogenase from higher plants involved in lignin biosynthesis. Phytochemistry. 19747 (1), 519-520.
- Protection of carboxylic acids as cinnamyl esters has been reported: J. Chem. Soc., Chem. Commun., 707 (1996). They can be cleaved under neutral conditions with Hg(II) or KSCN: Tetrahedron Lett., 2081 (1977).
- Allylic alcohols react with primary or secondary amines in the presence of SnCl2 and a Pd(0) catalyst to give allylic amines regioselectively: Chem. Lett., 1121 (1995):