Search
Thermo Scientific Chemicals
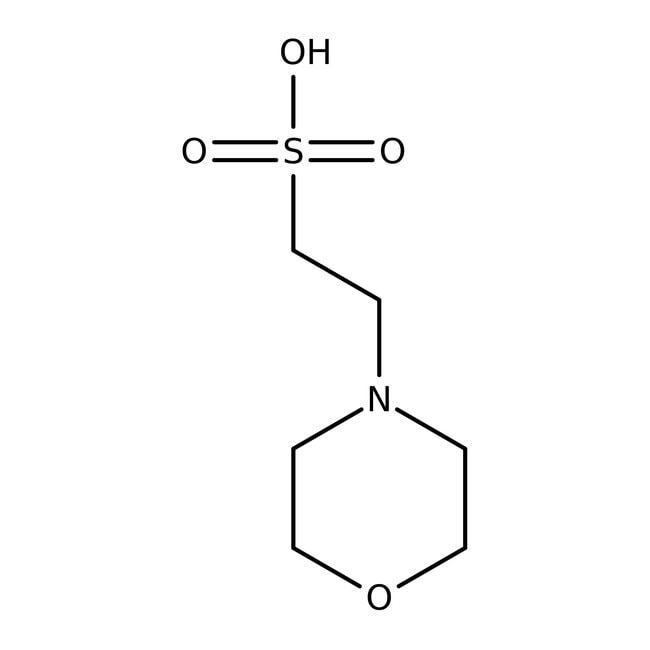
MES, 1.0M buffer soln., pH 6.5
CAS: 145224-94-8 | C6H15NO5S | 213.248 g/mol
Catalog number J61587.AP
also known as J61587-AP
Price (JPY)
-
Quantity:
500 mL
Chemical Identifiers
CAS145224-94-8
IUPAC Name4-(2-sulfonatoethyl)morpholin-4-ium
Molecular FormulaC6H13NO4S
InChI KeySXGZJKUKBWWHRA-UHFFFAOYSA-N
SMILES[O-]S(=O)(=O)CC[NH+]1CCOCC1
View more
Specifications Specification Sheet
Specification Sheet
CommentAll components are dissolved in UV treated 18.2 megohm-cm ± 1 water, followed by pH adjustment and filtered through 0.22 micron filter.
pH6.5 ± 0.20
FormLiquid
Appearance (Color)Clear colorless to pale yellow
MES is used as a buffering agent in biology and biochemistry. It is utilized for resolving very small proteins on bis-tris gels. It is mainly used as a biological buffer in plant cell cultures. Buffer solutions are used as a means of keeping pH at a nearly constant value in a wide variety of chemical applications.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
MES is used as a buffering agent in biology and biochemistry. It is utilized for resolving very small proteins on bis-tris gels. It is mainly used as a biological buffer in plant cell cultures. Buffer solutions are used as a means of keeping pH at a nearly constant value in a wide variety of chemical applications.
Solubility
Miscible with water.
Notes
Incompatible with strong oxidizing agents.
MES is used as a buffering agent in biology and biochemistry. It is utilized for resolving very small proteins on bis-tris gels. It is mainly used as a biological buffer in plant cell cultures. Buffer solutions are used as a means of keeping pH at a nearly constant value in a wide variety of chemical applications.
Solubility
Miscible with water.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Camargo, T. P.; Maia, F. F.; Chaves, C.; de Souza, B.; Bortoluzzi, A. J.; Castilho, N.; Bortolotto, T.; Terenzi, H.; Castellano, E. E.; Haase, W.; Tomkowicz, Z.; Peralta, R. A.; Neves, A. Synthesis, characterization, hydrolase and catecholase activity of a dinuclear iron (III) complex: Catalytic promiscuity. J. Inorg. Biochem. 2015, 146, 77-88.
- Rahman, A.; Raja, N. Z. R.; Ali, M.; Sugiyama, S.; TC Leow, A.; Inoue, Mahiran, B.; Abu, B. S.; T.; Matsumura, H. A Comparative Analysis of Microgravity and Earth Grown Thermostable T1 Lipase Crystals Using HDPCG Apparatus. Protein Pept. Lett. 2015, 22 (2), 173-179.