Search
Thermo Scientific Chemicals
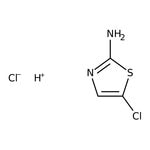
2-Amino-5-chlorothiazole hydrochloride, 97%
CAS: 55506-37-1 | C3H4Cl2N2S | 171.04 g/mol
Chemical Identifiers
CAS55506-37-1
IUPAC Namehydrogen 5-chloro-1,3-thiazol-2-amine chloride
Molecular FormulaC3H4Cl2N2S
InChI KeyGTMGFQYVLSQTKP-UHFFFAOYSA-N
SMILES[H+].[Cl-].NC1=NC=C(Cl)S1
View more
Specifications Specification Sheet
Specification Sheet
FormPowder or granules
Appearance (Color)Dark cream to cream to pale brown or pale pink to gray
Assay (Titration ex Chloride)≥96.0 to ≤104.0%
2-Amino-5-chlorothiazole hydrochloride is used as starting reagent in the synthesis of 2-chloro-6-methylimidazo[2,1-b]thiazole. It is used as reactant for preparation of biologically active thiazole derivatives.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Amino-5-chlorothiazole hydrochloride is used as starting reagent in the synthesis of 2-chloro-6-methylimidazo[2,1-b]thiazole. It is used as reactant for preparation of biologically active thiazole derivatives.
Solubility
Soluble in water.
Notes
Store in a cool, dry conditions in well sealed container. Incompatible with oxidizing agents.
2-Amino-5-chlorothiazole hydrochloride is used as starting reagent in the synthesis of 2-chloro-6-methylimidazo[2,1-b]thiazole. It is used as reactant for preparation of biologically active thiazole derivatives.
Solubility
Soluble in water.
Notes
Store in a cool, dry conditions in well sealed container. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Aldo Andreani; Massimiliano Granaiola; Alberto Leoni; Alessandra Locatelli; Rita Morigi; Mirella Rambaldi; Maurizio Recanatini; Giorgio Lenaz; Romana Fato; Christian Bergamini. Effects of new ubiquinone-imidazo[2,1-b]thiazoles on mitochondrial complex I (NADH-ubiquinone reductase) and on mitochondrial permeability transition pore. Bioorganic & Medicinal Chemistry. 2004, 12, (21), 5525-5532.
- K Tsuji; T Ogino; N Seki; M Sawada; Y Sudo; F Nishigaki; T Manda; M Matsuo. Synthesis and effects of novel thiazole derivatives against thrombocytopenia. Bioorganic & Medicinal Chemistry Letters. 1998, 8, (18), 2473-2478.