Search
Thermo Scientific Chemicals
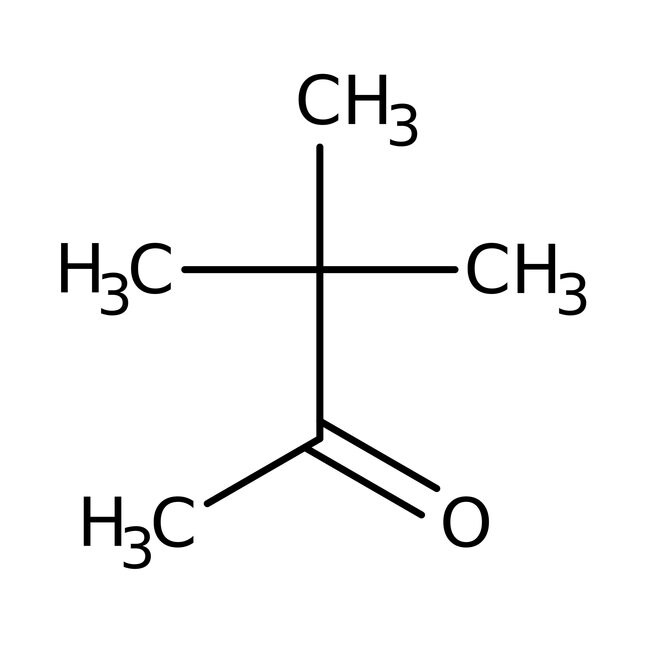
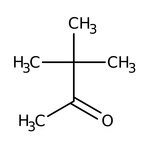
Pinacolone, 97%, Thermo Scientific Chemicals
Chemical Identifiers
CAS75-97-8
IUPAC Name3,3-dimethylbutan-2-one
Molecular FormulaC6H12O
InChI KeyPJGSXYOJTGTZAV-UHFFFAOYSA-N
SMILESCC(=O)C(C)(C)C
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear, colourless to yellow
Formliquid
Refractive Index1.3945 - 1.3995 @20?C
Assay (GC)> 96.0%
Pinacolone is a precursor to triazolylpinacolone, triadimefon, paclobutrazol, uniconazole and metribuzin. Furthermore, it acts as an intermediate for biologically active products such as antibacterial, antifungal, antiviral and antituberculous products. It is an important ketone in organic chemistry and participates in condensation, hydrogenation and reductive amination reactions.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Pinacolone is a precursor to triazolylpinacolone, triadimefon, paclobutrazol, uniconazole and metribuzin. Furthermore, it acts as an intermediate for biologically active products such as antibacterial, antifungal, antiviral and antituberculous products. It is an important ketone in organic chemistry and participates in condensation, hydrogenation and reductive amination reactions.
Solubility
Miscible with water, chloroform, ethanol, ether, acetone and carbon tetrachloride.
Notes
Incompatible with strong oxidizing agents.
Pinacolone is a precursor to triazolylpinacolone, triadimefon, paclobutrazol, uniconazole and metribuzin. Furthermore, it acts as an intermediate for biologically active products such as antibacterial, antifungal, antiviral and antituberculous products. It is an important ketone in organic chemistry and participates in condensation, hydrogenation and reductive amination reactions.
Solubility
Miscible with water, chloroform, ethanol, ether, acetone and carbon tetrachloride.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Gelat, F.; Richard, V.; Berger, O.; Montchamp, J. L. Development of a New Family of Chiral Auxiliaries. Org. Lett. 2015, 17 (8), 1819-1821.
- Braun, M.; Ito, T.; Hoshino, R.; Iinuma, M. Lithium Enolates:'Capricious' Structures-Reliable Reagents for Synthesis. Helv. Chim. Acta. 2015, 98 (1), 1-31.