Search
Thermo Scientific Chemicals
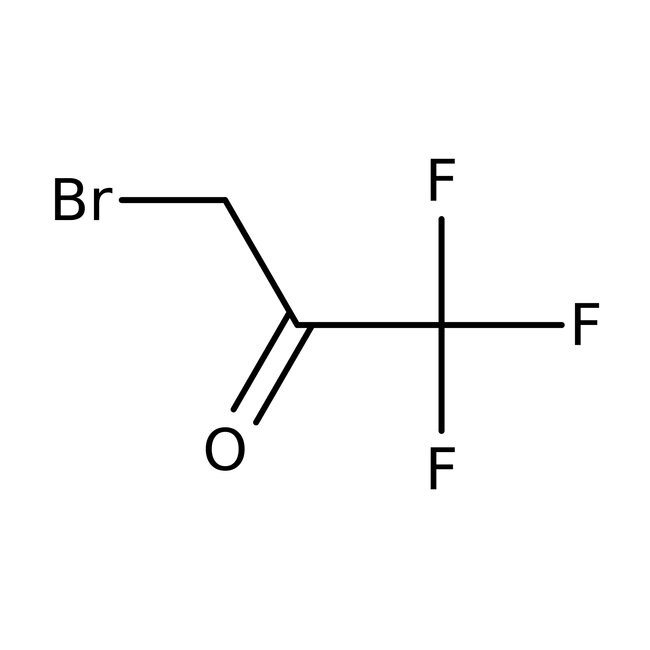
1-Bromo-3,3,3-trifluoroacetone, 97%
CAS: 431-35-6 | C3H2BrF3O | 190.95 g/mol
Catalog number A14948.14
also known as A14948-14
Price (JPY)
-
Quantity:
25 g
Chemical Identifiers
CAS431-35-6
IUPAC Name3-bromo-1,1,1-trifluoropropan-2-one
Molecular FormulaC3H2BrF3O
InChI KeyONZQYZKCUHFORE-UHFFFAOYSA-N
SMILESFC(F)(F)C(=O)CBr
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Assay (GC)≥96.0%
Appearance (Color)Clear colorless to yellow
Refractive Index1.3730-1.3780 @ 20?C
1-Bromo-3,3,3-trifluoroacetone is used in the preparation of 3-nonylthio-1,1,1-trifluoropropan-2-one and perfluoroalkylated trans-allylic alcohols. It is involved in the synthesis of cyclic tetrapeptides. Further, it reacts with potassium enolate of ethyl 4,4,4-trifluoroacetoacetate to produce ethyl 2,4-bis(trifluoromethyl)-4-hydroxydihydro-3-furoate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1-Bromo-3,3,3-trifluoroacetone is used in the preparation of 3-nonylthio-1,1,1-trifluoropropan-2-one and perfluoroalkylated trans-allylic alcohols. It is involved in the synthesis of cyclic tetrapeptides. Further, it reacts with potassium enolate of ethyl 4,4,4-trifluoroacetoacetate to produce ethyl 2,4-bis(trifluoromethyl)-4-hydroxydihydro-3-furoate.
Solubility
Immiscible with water.
Notes
Incompatible with strong oxidizing agents and bases.
1-Bromo-3,3,3-trifluoroacetone is used in the preparation of 3-nonylthio-1,1,1-trifluoropropan-2-one and perfluoroalkylated trans-allylic alcohols. It is involved in the synthesis of cyclic tetrapeptides. Further, it reacts with potassium enolate of ethyl 4,4,4-trifluoroacetoacetate to produce ethyl 2,4-bis(trifluoromethyl)-4-hydroxydihydro-3-furoate.
Solubility
Immiscible with water.
Notes
Incompatible with strong oxidizing agents and bases.
RUO – Research Use Only
General References:
- A novel reductive olefination reaction with aldehydes, mediated by Ti(O-i-Pr)4 and PPh3, provides a one-pot synthesis of trifluoromethylated trans-allylic alcohols: Chem. Commun., 2195 (1998):
- The oxime is readily converted with base to the highly reactive species 1,1,1-trifluoro-2-nitroso-2-propene, which undergoes hetero-Diels-Alder reactions, e.g. with cyclopentadiene or indole to give fused oxazolines. The indole adduct rearomatizes to give 1-(3-indolyl)-3,3,3-trifluoroacetoxime: J. Org. Chem., 57, 339 (1992).
- Hiraoka, Y.; Kawasaki-Takasuka, T.; Morizawa, Y.; Yamazaki, T. Synthetic utility of 2,3,3,3-tetrafluoroprop-1-ene (HFO-1234yf). J. Fluorine Chem. 2015, 179, 71-76.
- Dalmal, T.; Appalanaidu, K.; Kosurkar, U. B.; Jagadeesh Babu, N.; Kumbhare, R. M. One-Pot Synthesis of 2-Imino-4-(trifluoromethyl)thiazolidin-4-ol Derivatives in a Three-Component Reaction: Application to Structurally Diverse Scaffolds of Biological Interest Through Subsequent Reactions. Eur. J. Org. Chem. 2014, 2014 (12), 2468-2479.