Search
Thermo Scientific Chemicals
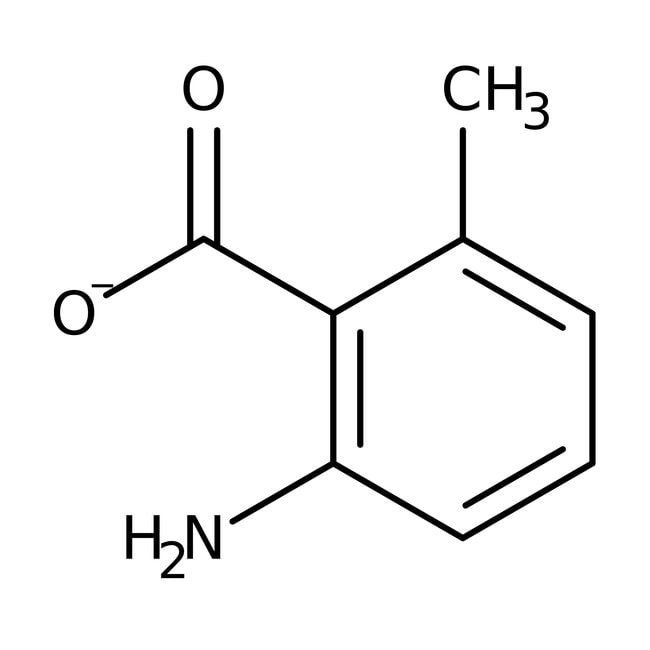
2-Amino-6-methylbenzoic acid, 98%
CAS: 4389-50-8 | C8H8NO2 | 150.16 g/mol
Catalog number A15187.22
also known as A15187-22
Price (JPY)
-
Quantity:
100 g
Chemical Identifiers
CAS4389-50-8
IUPAC Name2-amino-6-methylbenzoate
Molecular FormulaC8H8NO2
InChI KeyXHYVBIXKORFHFM-UHFFFAOYSA-M
SMILESCC1=CC=CC(N)=C1C([O-])=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Pale cream to cream to pale brown
FormPowder
Assay (Aqueous acid-base Titration)≥97.5 to ≤102.5%
Assay (Silylated GC)≥97.5%
2-Amino-6-methylbenzoic acid reacts with cyanogen bromide to prepare 2-amino-5-methyl-benzo[d][1,3]oxazin-4-one.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Amino-6-methylbenzoic acid reacts with cyanogen bromide to prepare 2-amino-5-methyl-benzo[d][1,3]oxazin-4-one.
Solubility
Soluble in methanol.
Notes
Incompatible with strong oxidizing agents and strong acids.
2-Amino-6-methylbenzoic acid reacts with cyanogen bromide to prepare 2-amino-5-methyl-benzo[d][1,3]oxazin-4-one.
Solubility
Soluble in methanol.
Notes
Incompatible with strong oxidizing agents and strong acids.
RUO – Research Use Only
General References:
- Ma, C.; Gong, G.; Liu, Z.; Ma, A.; Chen, Z. Stimulatory effects of tea supplements on the propagation of Lactobacillus casei in milk. Int. Dairy. J. 2015, 43, 1-6.
- Pearson, A. J.; Panda, S. N-Prolinylanthranilic acid derivatives as bifunctional organocatalysts for asymmetric aldol reactions. Tetrahedron 2011, 67 (22), 3969-3975.
- Wang, D.; Lin, H.; Cao, R.; Yang, S.; Chen, Q.; Hao, G.; Yang, W.; Yang, G. Synthesis and Herbicidal Evaluation of Triketone-Containing Quinazoline-2,4-diones. J. Agric. Food Chem. 2014, 62 (49), 11786-11796.