Search
Thermo Scientific Chemicals
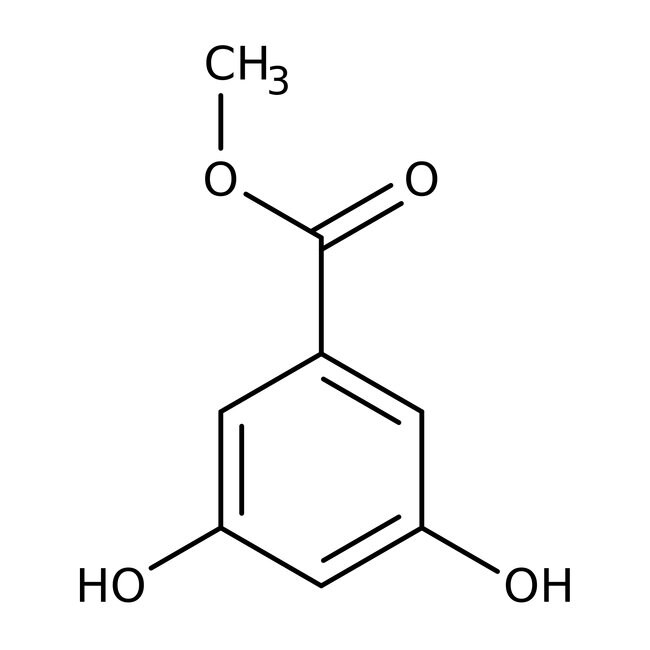
Methyl 3,5-dihydroxybenzoate, 98%
CAS: 2150-44-9 | C8H8O4 | 168.15 g/mol
Catalog number A15224.09
also known as A15224-09
Price (JPY)
-
Quantity:
10 g
Chemical Identifiers
CAS2150-44-9
IUPAC Namemethyl 3,5-dihydroxybenzoate
Molecular FormulaC8H8O4
InChI KeyRNVFYQUEEMZKLR-UHFFFAOYSA-N
SMILESCOC(=O)C1=CC(O)=CC(O)=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream or pale brown
FormCrystals or powder or crystalline powder
Assay (Silylated GC)≥97.5%
Assay (HPLC)≥97.5%
Melting Point (clear melt)164.0-170.0?C
Methyl 3,5-dihydroxybenzoate was used in the synthesis of cored dendrimers. It was also used in the preparation of bis(5-carbomethoxy-1,3-phenylene)-32-crown-10, a semi-rigid 32-membered ring diester crown ether. It has been used to study ribonucleotide reductase inhibiton and anti-tumor activity .
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Methyl 3,5-dihydroxybenzoate was used in the synthesis of cored dendrimers. It was also used in the preparation of bis(5-carbomethoxy-1,3-phenylene)-32-crown-10, a semi-rigid 32-membered ring diester crown ether. It has been used to study ribonucleotide reductase inhibiton and anti-tumor activity .
Solubility
Slightly soluble in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
Methyl 3,5-dihydroxybenzoate was used in the synthesis of cored dendrimers. It was also used in the preparation of bis(5-carbomethoxy-1,3-phenylene)-32-crown-10, a semi-rigid 32-membered ring diester crown ether. It has been used to study ribonucleotide reductase inhibiton and anti-tumor activity .
Solubility
Slightly soluble in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Michael S.Wendlan; Steven C.Zimmerman. Synthesis of cored dendrimers. J. Am. Chem. Soc. 1999, 121,(6), 1389-1390.
- Yadollah Delaviz; Harry W.Gibson. Macrocyclic polymers. 1. Synthesis of a poly(ester crown) based on bis(5-carboxy-1,3-phenylene)-32-crown-10 and 4,4'-isopropylidenediphenol (bisphenol A). Macromolecules. 1992, 25,(1), 18-20.