Search
Thermo Scientific Chemicals
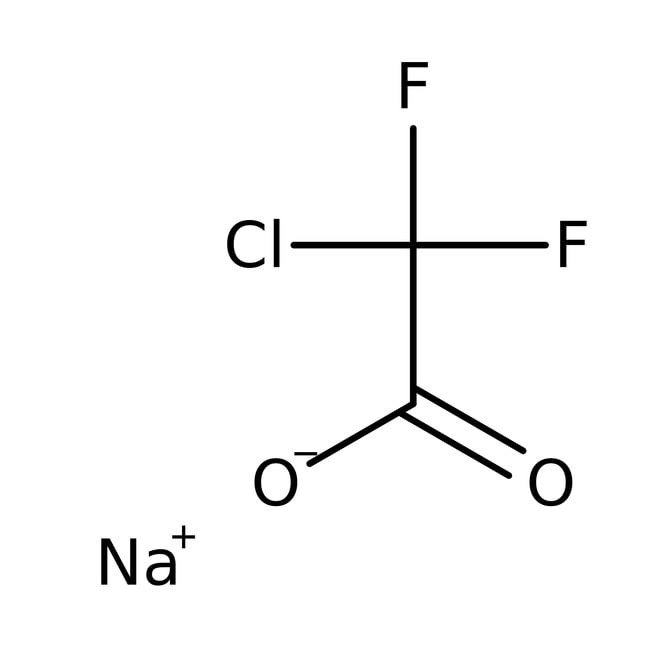
Sodium chlorodifluoroacetate, 97%
CAS: 1895-39-2 | C2ClF2NaO2 | 152.457 g/mol
Catalog number A15756.14
also known as A15756-14
Price (JPY)
-
Quantity:
25 g
Chemical Identifiers
CAS1895-39-2
IUPAC Namesodium 2-chloro-2,2-difluoroacetate
Molecular FormulaC2ClF2NaO2
InChI KeyMRTAVLDNYYEJHK-UHFFFAOYSA-M
SMILES[Na+].[O-]C(=O)C(F)(F)Cl
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream
FormCrystals or powder or crystalline powder
Assay (Non-aqueous acid-base Titration)≥96.0 to ≤104.0%
It is used as pharmaceutical intermediate. Reactions of aldehydes with triphenylphosphine and sodium chlorodifluoroacetate yield 1,1-difluoro olefins. It is a general reaction and a one step synthesis.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is used as pharmaceutical intermediate. Reactions of aldehydes with triphenylphosphine and sodium chlorodifluoroacetate yield 1,1-difluoro olefins. It is a general reaction and a one step synthesis.
Solubility
Soluble in dimethoxyethane, diglyme (0.60 g mL-1), triglyme, dimethyl formamide.
Notes
Store at room temperature. Keep away from oxidizing agents.
It is used as pharmaceutical intermediate. Reactions of aldehydes with triphenylphosphine and sodium chlorodifluoroacetate yield 1,1-difluoro olefins. It is a general reaction and a one step synthesis.
Solubility
Soluble in dimethoxyethane, diglyme (0.60 g mL-1), triglyme, dimethyl formamide.
Notes
Store at room temperature. Keep away from oxidizing agents.
RUO – Research Use Only
General References:
- Samuel A. Fuqua.; Warren G. Duncan.; Robert M. Silverstein.Synthesis of 1,1-Difluoro Olefins. II. Reactions of Ketones with Tributylphosphine and Sodium Chlorodifluoroacetate. J. Org. Chem. 1965, 30 (8),2543-2545 .
- Samuel A. Fuqua.; Warren G. Duncan.; Robert M. Silverstein.A One-Step Synthesis of 1,1-Difluoro Olefins from Aldehydes. J. Org. Chem. 1965, 30 (4),1027-1029 .
- Thermolysis in refluxing diglyme generates difluorocarbene: Proc. Chem. Soc., 81 (1960); Tetrahedron, 25, 1219 (1969); Tetrahedron Lett., 1319 (1973). In triglyme at 165°, gave better results in comparison with most other difluorocarbene precursors in cyclopropanation of an enol ether: Tetrahedron, 46, 5213 (1990).
- In the presence of triphenyl- or tributylphosphine, the carbene is intercepted to give the difluoromethylenephosphorane, which can be used to convert aldehydes and ketones to 1,1-difluoroalkenes: Tetrahedron Lett., 1461 (1964); J. Org. Chem., 30, 1027, 2543 (1965); Org. Synth. Coll., 5, 390, 949 (1973):