Search
Thermo Scientific Chemicals
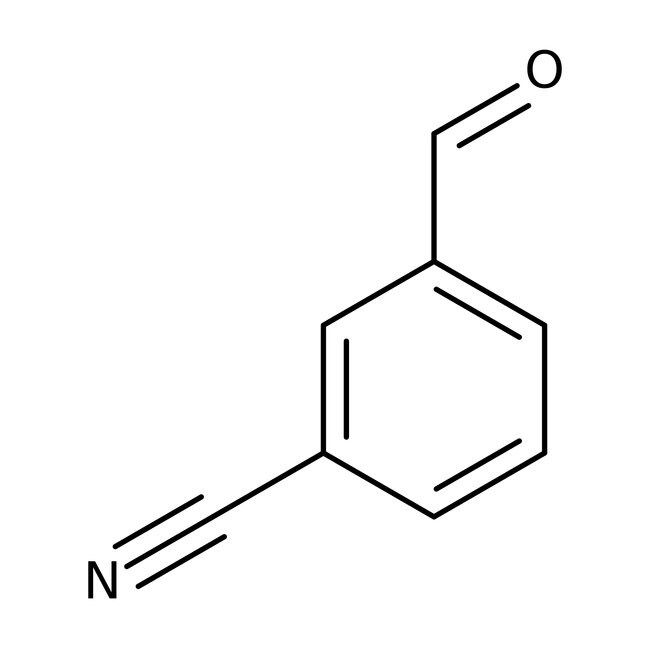
3-Cyanobenzaldehyde, 97%
CAS: 24964-64-5 | C8H5NO | 131.134 g/mol
Chemical Identifiers
CAS24964-64-5
IUPAC Name3-formylbenzonitrile
Molecular FormulaC8H5NO
InChI KeyHGZJJKZPPMFIBU-UHFFFAOYSA-N
SMILESO=CC1=CC=CC(=C1)C#N
View more
Specifications Specification Sheet
Specification Sheet
Melting Point (clear melt)73.0-82.0?C
Free acid (titration)≤1.5%
Assay (GC)≥96.0%
FormCrystals or powder or crystalline powder or granules
Appearance (Color)White to cream to yellow
3-Cyanobenzaldehyde is used in the synthesis of 3-(6,6-dimethyl-5,6-dihydro-4H-benzo[7,8]chromeno[6,5-d]oxazol-2-yl)benzonitrile. It is an important raw material and intermediate used in organic synthesis, pharmaceuticals agrochemicals and dyestuff fields.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Cyanobenzaldehyde is used in the synthesis of 3-(6,6-dimethyl-5,6-dihydro-4H-benzo[7,8]chromeno[6,5-d]oxazol-2-yl)benzonitrile. It is an important raw material and intermediate used in organic synthesis, pharmaceuticals agrochemicals and dyestuff fields.
Solubility
Insoluble in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents. It is sensitive to air. Store under dry inert gas.
3-Cyanobenzaldehyde is used in the synthesis of 3-(6,6-dimethyl-5,6-dihydro-4H-benzo[7,8]chromeno[6,5-d]oxazol-2-yl)benzonitrile. It is an important raw material and intermediate used in organic synthesis, pharmaceuticals agrochemicals and dyestuff fields.
Solubility
Insoluble in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents. It is sensitive to air. Store under dry inert gas.
RUO – Research Use Only
General References:
- Phillip A. Hopes.; Alexandra J. Parker.; Ian Patel. Development and Optimisation of an Unsymmetrical Hantzsch Reaction for Plant-Scale Manufacture. Org. Process Res. Dev. 2006, 10 (4), 808-813.
- Kelly C.G. Moura.; Paula F. Carneiro.; Maria do Carmo F.R. Pinto.; José A. da Silva.; Valéria R.S. Malta.; Carlos A. de Simone.; Gleiston G. Dias.; Guilherme A.M. Jardim.; Jéssica Cantos.; Tatiane S. Coelho.; Pedro E. Almeida da Silva.; Eufrânio N. da Silva. 1,3-Azoles fromortho-naphthoquinones: Synthesis of aryl substituted imidazoles and oxazoles and their potent activity againstMycobacterium tuberculosis. Bioorganic & Medicinal Chemistry. 2012, 20 (21), 6482-6488.