Search
Thermo Scientific Chemicals
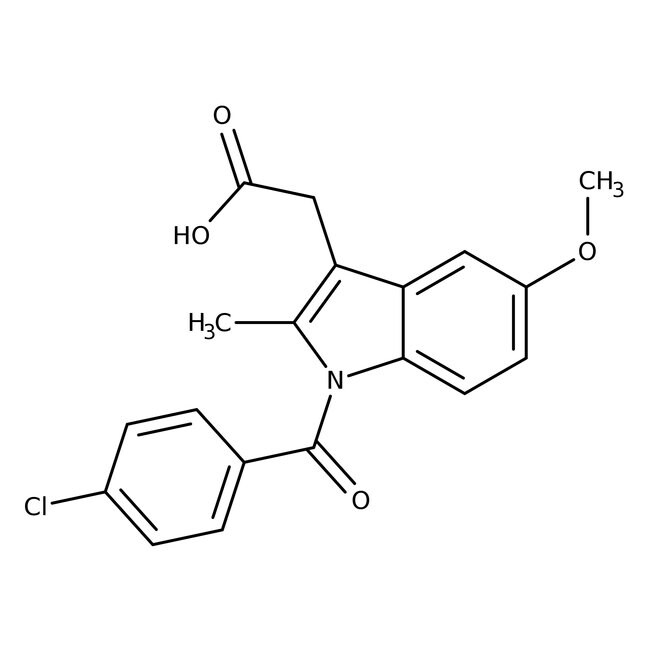
Indomethacin, 98%
CAS: 53-86-1 | C19H16ClNO4 | 357.79 g/mol
Chemical Identifiers
CAS53-86-1
IUPAC Name2-[1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indol-3-yl]acetic acid
Molecular FormulaC19H16ClNO4
InChI KeyCGIGDMFJXJATDK-UHFFFAOYSA-N
SMILESCOC1=CC=C2N(C(=O)C3=CC=C(Cl)C=C3)C(C)=C(CC(O)=O)C2=C1
View more
Specifications Specification Sheet
Specification Sheet
Assay (Aqueous acid-base Titration)≥97.5 to ≤102.5%
Melting Point (clear melt)156.0-163.0?C
FormPowder
Assay (HPLC)≥97.5%
Appearance (Color)White to pale cream
View more
It is a non-steroidal anti-inflammatory drug, it is used in the treatment of musculoskeletal and joint disorders including osteoarthritis, rheumatoid arthritis, gout, bursitis and tendinitis. It finds its application as gout suppressant, non-steroidal anti-inflammatory drug and analgesic. It also plays many biological roles. Additionally, Indomethacin displays adipogenic actions via binding and activation of PPARγ (peroxisome proliferator-activated receptor γ), a ligand-activated transcription factor known to play an important role in adipogenesis.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Solubility
Soluble in acetone (40 mg/mL - clear, yellow solution), ethanol (20 mg/mL), ether, castor oil; Soluble in chloroform (50 mg/mL - clear, yellow, extremely viscous solution); decomposed by strong alkali but stable in neutral or slightly acidic media; insoluble in water.
Notes
Store away from oxidizing agents, strong bases and light. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
Soluble in acetone (40 mg/mL - clear, yellow solution), ethanol (20 mg/mL), ether, castor oil; Soluble in chloroform (50 mg/mL - clear, yellow, extremely viscous solution); decomposed by strong alkali but stable in neutral or slightly acidic media; insoluble in water.
Notes
Store away from oxidizing agents, strong bases and light. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
RUO – Research Use Only
General References:
- Tinny T.; Chacko AJ.; Jose S. Formulation development and statistical optimization of chronotherapeutic tablets of indometacin.Drug Dev. Ind. Pharm.2013,39(9), 1357-1363.
- Han L, et al. Indometacin ameliorates high glucose-induced proliferation and invasion via modulation of e-cadherin in pancreatic cancer cells. Current medicinal chemistry.2013,20(33), 4142-4152.