Search
Thermo Scientific Chemicals
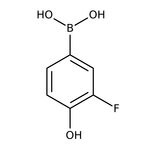
3-Fluoro-4-hydroxybenzeneboronic acid, 97%
CAS: 182344-14-5 | C6H6BFO3 | 155.919 g/mol
Chemical Identifiers
CAS182344-14-5
IUPAC Name(3-fluoro-4-hydroxyphenyl)boronic acid
Molecular FormulaC6H6BFO3
InChI KeyOYNDLOJPYURCJG-UHFFFAOYSA-N
SMILESOB(O)C1=CC(F)=C(O)C=C1
View more
Specifications Specification Sheet
Specification Sheet
Assay (HPLC)>96.0%
3-Fluoro-4-hydroxybenzeneboronic acid is used as intermediates. Hydroxybenzene boronic acids are involved in suzuki miyaura reactions. It is a arylboronic acid. 3-Fluoro-4-hydroxybenzeneboronic acid, can be an effective catalyst for amidation and esterification of carboxylic acids.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Fluoro-4-hydroxybenzeneboronic acid is used as intermediates. Hydroxybenzene boronic acids are involved in suzuki miyaura reactions. It is a arylboronic acid. 3-Fluoro-4-hydroxybenzeneboronic acid, can be an effective catalyst for amidation and esterification of carboxylic acids.
Solubility
Slightly soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
3-Fluoro-4-hydroxybenzeneboronic acid is used as intermediates. Hydroxybenzene boronic acids are involved in suzuki miyaura reactions. It is a arylboronic acid. 3-Fluoro-4-hydroxybenzeneboronic acid, can be an effective catalyst for amidation and esterification of carboxylic acids.
Solubility
Slightly soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Tomohiro Yasukawa et. al.Copper-catalyzed, aerobic oxidative cross-coupling of alkynes with arylboronic acids: remarkable selectivity in 2,6-lutidine media.. Organic & Biomolecular Chemistry. 2011, 9(18), 6208-6210.