Search
Thermo Scientific Chemicals
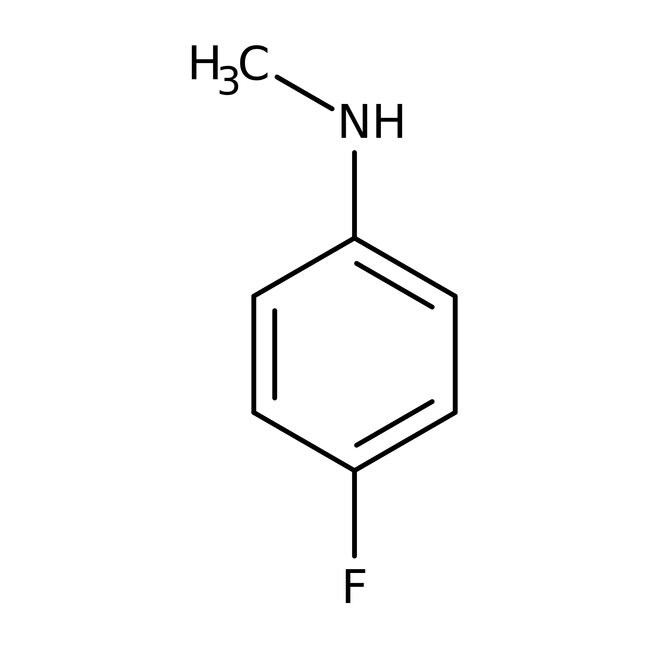
4-Fluoro-N-methylaniline, 97%
CAS: 459-59-6 | C7H8FN | 125.146 g/mol
Catalog number H55105.03
also known as H55105-03
Price (JPY)Request A Quote
-
Quantity:
1 g
Chemical Identifiers
CAS459-59-6
IUPAC Name4-fluoro-N-methylaniline
Molecular FormulaC7H8FN
InChI KeyVLWRKVBQUANIGI-UHFFFAOYSA-N
SMILESCNC1=CC=C(F)C=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Colorless to yellow to green
FormLiquid
Assay from Suppliers CofA≥96.0% (GC)
4-Fluoro-N-methylaniline is used as a model compound to study the biotransformation of secondary aromatic amines.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Fluoro-N-methylaniline is used as a model compound to study the biotransformation of secondary aromatic amines.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents , acids, acid chloride, acid anhydride and chloroformates.
4-Fluoro-N-methylaniline is used as a model compound to study the biotransformation of secondary aromatic amines.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents , acids, acid chloride, acid anhydride and chloroformates.
RUO – Research Use Only
General References:
- James P. Driscoll.; Ignacio Aliagas.; Jennifer J. Harris, Jason S. Halladay.; Sheerin Khatib-Shahidi.; Alan Deese.; Nathaniel Segraves.; S. Cyrus Khojasteh-Bakht. Formation of a Quinoneimine Intermediate of 4-Fluoro-N-methylaniline by FMO1: Carbon Oxidation Plus Defluorination. Chem. Res. Toxico. 2011, 23 (5), 861-863.
- M G Boersma.; N H Cnubben.; W J van Berkel.; M Blom.; J Vervoor.; I M Rietjens. Role of cytochromes P-450 and flavin-containing monooxygenase in the biotransformation of 4-fluoro-N-methylaniline. DMD. 1993, 21 (2), 218-230.