Search
Thermo Scientific Chemicals
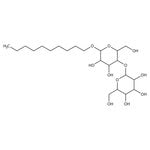
Decyl beta-D-maltopyranoside, 97+%
CAS: 82494-09-5 | C22H42O11 | 482.567 g/mol
Chemical Identifiers
CAS82494-09-5
IUPAC Name2-{[6-(decyloxy)-4,5-dihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy}-6-(hydroxymethyl)oxane-3,4,5-triol
Molecular FormulaC22H42O11
InChI KeyWOQQAWHSKSSAGF-UHFFFAOYNA-N
SMILESCCCCCCCCCCOC1OC(CO)C(OC2OC(CO)C(O)C(O)C2O)C(O)C1O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream or pale yellow
FormCrystals or powder or crystalline powder
Assay from Suppliers CofA≥97.0%
Decyl beta-D-maltopyranoside is used in a study to assess α-helical membrane protein crystallization. It has also been used in a study to investigate enantiomer separation of drugs by micellar electrokinetic chromatography. It aids in solubilizing the active cytochrome oxidase from mitochondria.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Decyl beta-D-maltopyranoside is used in a study to assess α-helical membrane protein crystallization. It has also been used in a study to investigate enantiomer separation of drugs by micellar electrokinetic chromatography. It aids in solubilizing the active cytochrome oxidase from mitochondria.
Solubility
Soluble in methanol at 50mg/ml
Notes
Store at -20°C. Store away from oxidizing agents. Store in a cool, dry conditions in a well sealed container.
Decyl beta-D-maltopyranoside is used in a study to assess α-helical membrane protein crystallization. It has also been used in a study to investigate enantiomer separation of drugs by micellar electrokinetic chromatography. It aids in solubilizing the active cytochrome oxidase from mitochondria.
Solubility
Soluble in methanol at 50mg/ml
Notes
Store at -20°C. Store away from oxidizing agents. Store in a cool, dry conditions in a well sealed container.
RUO – Research Use Only
General References:
- Simon Newstead; Sébastien Ferrandon; So Iwata. Rationalizing alpha-helical membrane protein crystallization. Protein Science.2008, 17, (3), 466-472.
- K Otsuka; S Terabe. Enantiomer separation of drugs by micellar electrokinetic chromatography using chiral surfactants. Journal of Chromatography A.2000, 875, (1-2), 163-178.