Search
Thermo Scientific Chemicals
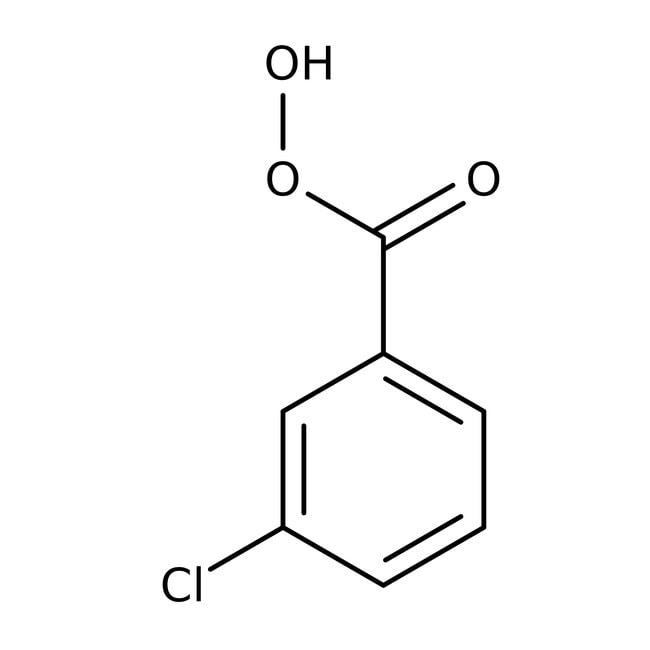
3-Chloroperoxybenzoic acid, 50-55%, cont. ca 10% 3-chlorobenzoic acid, balance water
CAS: 937-14-4 | C7H5ClO3 | 172.56 g/mol
| Catalog Number | Quantity |
|---|---|
| L00286.36 also known as L00286-36 | 500 g |
Catalog number L00286.36
also known as L00286-36
Price (JPY)
-
Quantity:
500 g
Chemical Identifiers
CAS937-14-4
IUPAC Name3-chlorobenzene-1-carboperoxoic acid
Molecular FormulaC7H5ClO3
InChI KeyNHQDETIJWKXCTC-UHFFFAOYSA-N
SMILESOOC(=O)C1=CC=CC(Cl)=C1
View more
Specifications Specification Sheet
Specification Sheet
Assay (Iodometric Titration)48.0-55.0%
FormMoist solid
Appearance (Color)White
Identification (FTIR)Conforms
3-Chloroperoxybenzoic acid is a strong oxidizing agent used in the oxidation reactions such as aldehydes and ketones to esters (Bayer-Villiger-Oxidation), olefines to epoxides, sulfides to sulfoxides and sulfones, and amines to nitroalkanes, N-oxides.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Chloroperoxybenzoic acid is a strong oxidizing agent used in the oxidation reactions such as aldehydes and ketones to esters (Bayer-Villiger-Oxidation), olefines to epoxides, sulfides to sulfoxides and sulfones, and amines to nitroalkanes, N-oxides.
Solubility
Soluble in potassium hydroxide, chloroform and acetone. Insoluble in water.
Notes
Heat sensitive. Incompatible with reactive with reducing agents, combustible materials, organic materials and alkalies.
3-Chloroperoxybenzoic acid is a strong oxidizing agent used in the oxidation reactions such as aldehydes and ketones to esters (Bayer-Villiger-Oxidation), olefines to epoxides, sulfides to sulfoxides and sulfones, and amines to nitroalkanes, N-oxides.
Solubility
Soluble in potassium hydroxide, chloroform and acetone. Insoluble in water.
Notes
Heat sensitive. Incompatible with reactive with reducing agents, combustible materials, organic materials and alkalies.
RUO – Research Use Only
General References:
- Reagent for epoxidation of alkenes: J. Org. Chem., 29, 1976 (1964). Dichloromethane has been recommended as solvent for these reactions, since the peracid is soluble, but the by-product, 3-chlorobenzoic acid, is almost insoluble. For high-yield, stereospecific epoxidation in a two-phase, almost neutral system, see: J. Org. Chem., 44, 1351 (1979). For regioselective epoxidation of the more substituted double bond of a diene, see: Org. Synth. Coll., 5, 467 (1973). For epoxidation in aqueous solution followed by in situ hydrolysis to the trans-diol by 10% sulfuric acid, see: Synth. Commun., 19, 1939 (1989).
- Carbonyl compounds undergo the Baeyer-Villiger reaction to give esters or lactones: J. Org. Chem., 29, 2914 (1964). For an extensive review of the Baeyer-Villiger reaction, see: Org. React., 43, 251 (1993).
- Primary aliphatic amines are oxidized to nitro-compounds: J. Org. Chem., 31, 524 (1966). Good yields are obtained in 1,2-dichloroethane solution: J. Org. Chem., 44, 659 (1979); 54, 2869 (1989).
- Sulfides can be oxidized selectively either to sulfoxides or sulfones: Tetrahedron, 22, 1235 (1966); J. Chem. Soc. (C), 2720 (1969); J. Org. Chem., 35, 2106 (1970).
- For the stereospecific oxidation of imines to oxaziridines under phase-transfer conditions, see: Org. Synth. Coll., 8, 546 (1993):
- Perfluorooxaziridines have been prepared using acetonitrile as solvent: J. Org. Chem., 58, 4754 (1993).
- For a brief feature on uses of this reagent in Organic synthesis, see: Synlett, 664 (2007).
- WARNING: The reagent has been reported to react exothermically with DMF: Org. Process Res. Dev., 6, 159 (2004).