Search
Thermo Scientific Chemicals
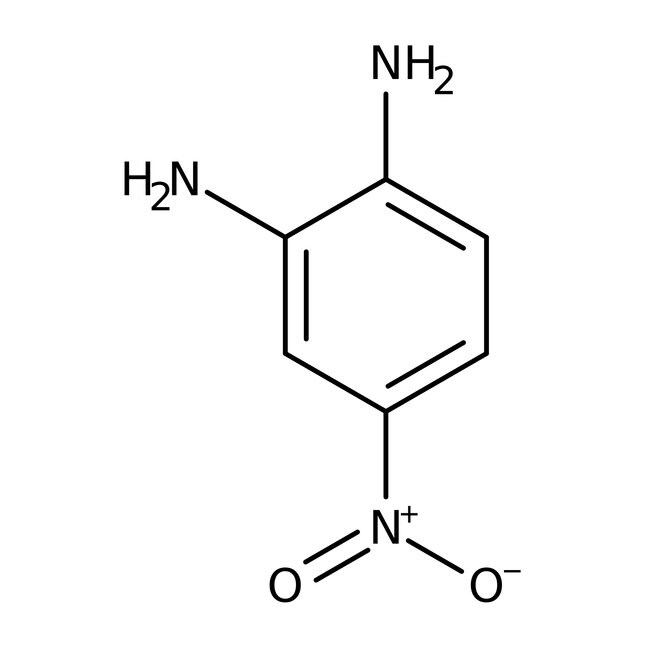
4-Nitro-o-phenylenediamine, 97%
CAS: 99-56-9 | C6H7N3O2 | 153.14 g/mol
Catalog number L04693.36
also known as L04693-36
Price (JPY)
-
Quantity:
500 g
Chemical Identifiers
CAS99-56-9
IUPAC Name4-nitrobenzene-1,2-diamine
Molecular FormulaC6H7N3O2
InChI KeyRAUWPNXIALNKQM-UHFFFAOYSA-N
SMILESNC1=CC=C(C=C1N)[N+]([O-])=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Orange to red to dark red or red-brown to brown
Identification (FTIR)Conforms
Assay (Non-aqueous acid-base Titration)≥96.0 to ≤104.0%
Melting Point (clear melt)199-207?C
FormPowder
View more
4-Nitro-o-phenylenediamine is used to study the in vitro activation of 4-nitro-o-phenylenediamine into a potent mutagen in Salmonella typhimurium strain TA98
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Nitro-o-phenylenediamine is used to study the in vitro activation of 4-nitro-o-phenylenediamine into a potent mutagen in Salmonella typhimurium strain TA98
Solubility
Slightly soluble in water (1.2 g/L at 20°C).
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
4-Nitro-o-phenylenediamine is used to study the in vitro activation of 4-nitro-o-phenylenediamine into a potent mutagen in Salmonella typhimurium strain TA98
Solubility
Slightly soluble in water (1.2 g/L at 20°C).
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Motoharu Tanaka.; Takuji Kawashima. Some 4-substituted o-phenylenediamines as reagents for selenium. Talanta. 1965, 12 (3), 211-219.
- N. N. Kolos.; V. D. Orlov.; E. Ya. Yuzefovskaya.; F. G. Yaremenko.; N. P. Vorob'eva.; O. V. Shishkin.; Yu. T. Struchkov.; S. M. Ivkov. Synthesis of aromatic derivatives of 1,5-benzodiazepine in the reaction of 4-nitro-o-phenylenediamine with chalcone dibromides. Chemistry of Heterocyclic Compounds. 1995, 31 (7), 827-834.