Search
QuantStudio™ 6 and QuantStudio™ 7 Flex Real-Time PCR Systems Support – Getting Started
Find valuable information.
Optimize your experiments to get the best results. We’ve compiled a detailed knowledgebase of the top tips and tricks to meet your research needs.
View the relevant questions below:
Having problems with your experiment? Visit our
| Recommended Maintenance Schedule | |
|---|---|
| Power on/off the computer controlling the instrument | Weekly |
| Check computer disk space. If necessary, archive or back up your experiment files and instrument settings | Weekly |
| Background calibration | Every month |
| Run disk cleanup and disk defragmentation | Every month |
| Perform an instrument self test | Every month |
| Pure dye calibrations | Every 6 months |
| ROI calibration | Every 6 months |
| Uniformity calibration | Every 6 months |
| Normalization calibration | Every 6 months |
| RNaseP instrument verification | After the instrument has been moved, or as needed to verify instrument performance |
The uniformity calibration generates data that allows the software to compensate for the physical effects of the QuantStudio™ system filters using the ROI (Region of Interest) plate. The normalization calibration generates factors that the software uses when comparing data from multiple QuantStudio™ instruments within a study. There are 2 normalization plates provided in the calibration kit, one containing FAM™/ROX™ dye and the other with VIC®/ROX™ dye.
All the calibration plates can be stored and reused three times for up to 6 months after you first open them, so make sure to return them to their original packaging return them to –20°C storage until the next use. (Note: Due to the small volumes, calibration array cards cannot be reused. Please discard them after use.) If needed, you can make your own background plate using deionized water. Please follow the directions in the QuantStudio™ 6 and 7 Flex Real-Time PCR Systems Maintenance and Administration Guide (Appendix C) for more details.
Please ask your dye manufacturer for recommendations. If there is no specific recommendation available, try TE Buffer (pH 8.0) to start.
The RNaseP verification plate contains template, master mix, and a TaqMan® assay for RNaseP. It is used to verify that the instrument is performing to specifications. If you have reason to suspect there is something wrong with the instrument, or if you want to rule out a chemistry issue,the RNaseP plate is a good way to test the system. The RNaseP verification plate is a single-use plate.
Please see the comparison table in this brochure for details on the differences between the two instruments.
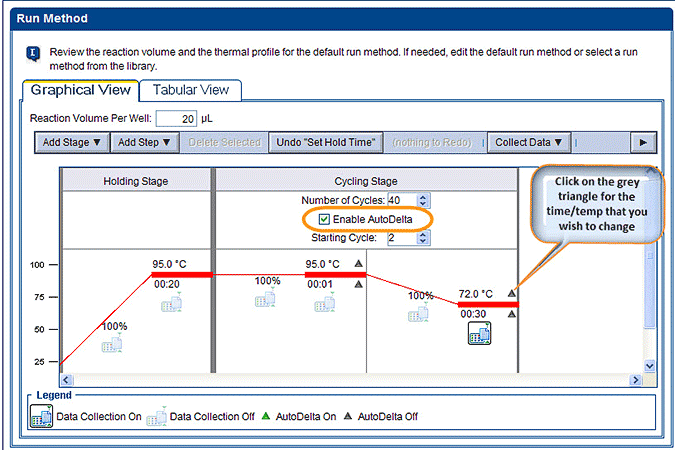
In a touchdown PCR experiment, you will change either the temperature or the time of a particular PCR step with every cycle. Most commonly, the annealing temperature is adjusted throughout the experiment, such that the specificity is higher in the early cycles and the efficiency in the later cycles. In this example, we will set the method to do the following:
40 cycles  |
|
- Go to File → New Experiment → Advanced Setup. Fill out the relevant options as you normally would.
- Go to the Run Method under the ‘Setup’ section and you should see the Graphical View of your thermal profile. Check the box next to ‘Enable AutoDelta’. You should see some grey triangles appear next to the Temperature and Time at every step in the Cycling Stage. (Note: if you want to start the changes at a later cycle set this here under ‘Starting Cycles’.).
- A new window called ‘AutoDelta Settings’ will open up. Select the appropriate options. In this example we are decreasing the temperature by 0.4°C per cycle, so choose (“-“) and (0.40). Click ‘Save Setting’. You will then see a green triangle show up next to the parameter you changed, in this case next to the 72°C step. Your new method has now been applied.

You will need an oligo sequence with the custom dye but without the quencher molecule. For each custom dye, prepare samples in a concentration range of 100–2,000nM, choosing a 2–3 fold difference in dilution points. Use either the background buffer provided in the calibration kit or your own buffer to dilute the oligo. A full plate is not needed; see the example below.

In this example, sample concentrations would be 100, 200, 400, 800, and 1,600 (20 µL/well).
Note: The volume would be the same for a 96-well or 384-well plate.
Set up a ‘dummy’ run using the ‘Standard Curve’ option. Alter the thermal profile so that it simply ramps to 60°C with a 2 min hold. Ensure that the filters of interest are selected.
When the run is complete, export and examine the raw data. Select the concentration to use by finding the dilution that will give you an acceptable signal in the following ranges:
- For a 384-well plate: between 400,000 and 1,200,000
- For a 96-well plate: between 1,400,000 and 4,300,000
Create a full plate of the dye using the selected concentration and run the custom-dye calibration as normal, using 20 µL. (Refer to the QuantStudio™ 6 and 7 Flex Maintenance and Administration Guide, Appendix C, page 185, “Creating a custom dye plate”, for full details).
The QuantStudio™ 6 and QuantStudio™ 7 Flex Real-Time PCR Systems can store up to 100 runs on the instrument itself.
The following volumes are supported for each instrument block:
- -96-well standard (10–100 µL reactions)
- -96-well Fast (10–30 µL reactions)
- -384-well (5–20 µL reactions)
- -TaqMan® Array Micro Fluidic Cards (~1 µL reactions) – for QuantStudio™ 7 Flex Real-Time PCR System only
Yes, the QuantStudio™ 6 and QuantStudio™ 7 Flex Real-Time PCR Systems can collect data at multiple steps in the amplification stage. You will need to turn on the data collection for every step of interest. In the analysis, only one set of data can be displayed at a time. To change the data set used for analysis, go to Analysis Settings → Ct Settings and change the drop down under ‘Data Step Selection’.
Run files will be saved to both the instrument and the connected computer. On the connected computer, files will be saved to the default data folder, unless you change it.
To find or change the default folder, go to → Tools → Preferences → Defaults. Here you will see a Data Folder and an Import Folder. The default location is shown. If you want files to be saved to (or open from) a different location, click ‘Browse’, and choose the new folder.
Follow the directions in this blog post to transfer your license to another computer. (Please note that the instructions here still apply, although they are written for the QuantStudio™ 12K Flex Real-Time PCR System.)
MicroAmp 96-Well Tray for VeriFlex Blocks (Cat. No. 4379983) is needed to load 1 or 2 MicroAmp Optical 8-Tube Strips with Attached Optical Caps on the ProFlex, SimpliAmp, MiniAmp, MiniAmp Plus, and Veriti Thermal Cycler 96-well blocks, and QuantStudio 3 and QuantStudio 5 blocks.
The MicroAmp Optical 8-Tube Strip with Attached Optical Caps is compatible with the following instruments:
- ProFlex 3 x 32-well PCR System
- ProFlex 96-well PCR System
- Veriti 96-well Thermal Cycler
- SimpliAmp Thermal Cycler
- MiniAmp Thermal Cycler
- MiniAmp Plus Thermal Cycler
- 2720 Thermal Cycler
- GeneAmp PCR System 9700, 96-well
- 7000 PCR System
- 7300 Real-Time PCR System
- 7500 Real-Time PCR system
- QuantStudio Real-Time PCR Systems
- ViiA7 Real-Time PCR System, 96-well
The MicroAmp 8-Tube Strip with Attached Domed Caps is compatible with the following instruments:
- ProFlex 3 x 32-well PCR System
- ProFlex 96-well
- Veriti 96-well Thermal Cycler
- SimpliAmp Thermal Cycler
- MiniAmp Thermal Cycler
- MiniAmp Plus Thermal Cycler
- 2720 Thermal Cycler
- GeneAmp PCR System 9700, 96-well
MicroAmp 96-Well Tray for VeriFlex Blocks (Cat. No. 4379983) is needed to load 1 or 2 MicroAmp Optical 8-Tube Strips with Attached Domed Caps on the ProFlex, SimpliAmp, MiniAmp, MiniAmp Plus, and Veriti Thermal Cycler 96-well blocks.
Yes. The tubes are DNase/RNase/Human DNA free, as they are manufactured in a Class 100K ISO certified clean room UK production facility.
Yes. The tubes are DNase/RNase/Human DNA free, as they are manufactured in a Class 100K ISO certified clean room UK production facility.
These new strips have new features including a graduated 20 μL measuring mark for visual verification and dual end tabs for better labeling and handling. In addition, the individual caps help prevent cross contamination and reduce sample evaporation.
These new strips have new features including a graduated 20 μL measuring mark for visual verification and dual end tabs for better labeling and handling. In addition, the individual caps help prevent cross contamination and reduce sample evaporation.
Please refer to the table on Page 2 of this flyer for compatibility information.
Please refer to the table on Page 2 of this flyer for compatibility information.
Yes. The tubes are DNase/RNase/Human DNA free, as they are manufactured in a Class 100K ISO certified clean room UK production facility.
Yes. The tubes are DNase/RNase/Human DNA free, as they are manufactured in a Class 100K ISO certified clean room UK production facility.
These new strips have new features including a graduated 20 μL measuring mark for visual verification and dual end tabs for better labeling and handling. In addition, the individual caps help prevent cross contamination and reduce sample evaporation.
These new strips have new features including a graduated 20 μL measuring mark for visual verification and dual end tabs for better labeling and handling. In addition, the individual caps help prevent cross contamination and reduce sample evaporation.
Data Analysis
For genotyping data, we recommend TaqMan® Genotyper software. For relative quantitation, we recommend ExpressionSuite™ software.
The ‘reveal traces’ feature for genotyping runs allows you to trace the clusters throughout the PCR process. For some assays you may find better cluster discrimination at, say, cycle 40, as opposed to cycle 50. To use this feature, the amplification data needs to have been collected for the run.
- Go to ‘Analysis Settings’
- Under the default ‘Call Settings’ tab, Choose ‘Analyze Real-Time Rn Data’
- Click ‘Apply Analysis Settings’
- Under the Allelic Discrimination Plot, check the box next to ‘Reveal Traces’. You can then move the slide bar to see how the data changes with the cycles number.
In the example below, the image on the left shows the data at a full 40 cycles. The image on the right is the same data after revealing traces (grey lines). The clusters can be traced back by moving the cycle bar. Notice that one point is no longer called at the earlier cycle set point.

For Research Use Only. Not for use in diagnostic procedures.