Search
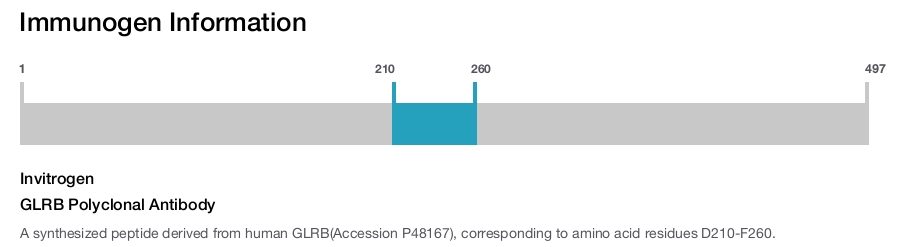
Invitrogen
GLRB Polyclonal Antibody
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
FIGURE: 1 / 2
GLRB Antibody (PA5-101610) in IHC (P)


Product Details
PA5-101610
Species Reactivity
Host/Isotype
Class
Type
Immunogen
Conjugate
Form
Concentration
Amount
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Product Specific Information
Antibody detects endogenous levels of total GLRB.
Target Information
In the central nervous system (CNS), glycine-mediated inhibitory neurotransmission is essential to voluntary motor control and reflex responses. Glycine binds to glycine receptors (GlyR) in the postsynaptic neuronal membranes. GlyR, gamma-aminobutryic acid, serotonin and acetylcholine comprise an evolutionally conserved superfamily of ligand-gated ion channels. The pentameric subunit structure of GlyR consists of two types of glycosylated membrane proteins, alpha1 through alpha4 and beta, and an associated peripheral membrane protein, which combine to form a chloride-selective ion channel. In humans, the composition of the pentamer changes from alpha2 subunits in the fetal CNS to alpha1 and beta subunits in the adult CNS. Fast potentiation of GlyR by intracellular Ca2+ in the brainstem and midbrain indicate an important role for Ca2+ in modulation of glycinergic synapses.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
References (0)
Bioinformatics
Protein Aliases: glycine receptor; Glycine receptor 58 kDa subunit; Glycine receptor subunit beta; glycine receptor, beta subunit; GlyR beta; inhibitory glycine receptor; ligand gated ion channel subunit; unnamed protein product
Gene Aliases: AI853901; GLRB; Glyrb; HKPX2; spa; spastic
UniProt ID: (Human) P48167, (Mouse) P48168, (Rat) P20781
Entrez Gene ID: (Human) 2743, (Mouse) 14658, (Rat) 25456

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support