Search
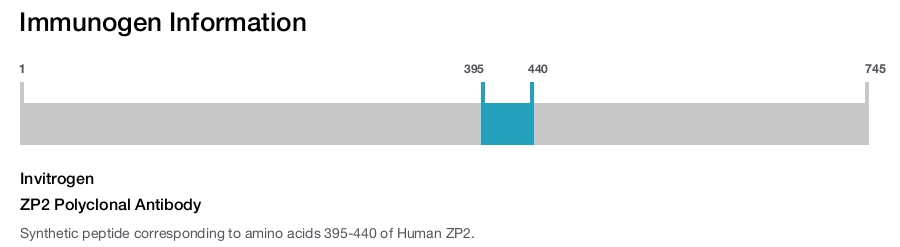
Invitrogen
ZP2 Polyclonal Antibody
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
FIGURE: 1 / 1
ZP2 Antibody (PA5-75949) in WB

Product Details
PA5-75949
Species Reactivity
Host/Isotype
Class
Type
Immunogen
Conjugate
Form
Concentration
Amount
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Product Specific Information
The antibody was affinity-purified from rabbit antiserum by affinity-chromatography using epitope-specific immunogen and the purity is > 95% (by SDS-PAGE).
Target Information
The zona pellucida is an extracellular matrix that surrounds the oocyte and early embryo. It is composed primarily of three or four glycoproteins with various functions during fertilization and preimplantation development. The protein encoded by this gene is a structural component of the zona pellucida and functions in secondary binding and penetration of acrosome-reacted spermatozoa. The nascent protein contains a N-terminal signal peptide sequence, a conserved ZP domain, a consensus furin cleavage site, and a C-terminal transmembrane domain. It is hypothesized that furin cleavage results in release of the mature protein from the plasma membrane for subsequent incorporation into the zona pellucida matrix. However, the requirement for furin cleavage in this process remains controversial based on mouse studies.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
References (0)
Bioinformatics
Protein Aliases: unnamed protein product; zona pellucida 2; zona pellucida 2 glycoprotein; Zona pellucida glycoprotein 2; zona pellucida glycoprotein 2 (sperm receptor); zona pellucida glycoprotein ZP2; Zona pellucida protein A; Zona pellucida sperm-binding protein 2; Zp-2
Gene Aliases: OOMD6; OZEMA6; Zp-2; ZP2; ZPA
UniProt ID: (Human) Q05996, (Rat) O54767, (Mouse) P20239
Entrez Gene ID: (Human) 7783, (Rat) 81828, (Mouse) 22787

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support