Search
Thermo Scientific Chemicals
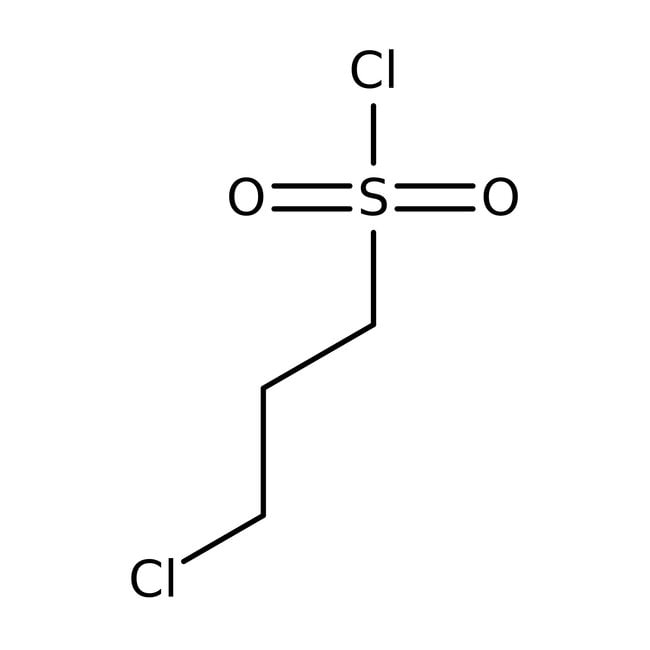
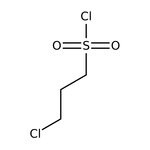
3-Chloropropanesulfonyl chloride, 98+%
CAS: 1633-82-5 | C3H6Cl2O2S | 177.039 g/mol
化学物質識別子
CAS1633-82-5
IUPAC Name3-chloropropane-1-sulfonyl chloride
Molecular FormulaC3H6Cl2O2S
InChI KeyGPKDGVXBXQTHRY-UHFFFAOYSA-N
SMILESClCCCS(Cl)(=O)=O
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear colorless to yellow to orange or pale brown
FormLiquid
Assay (GC)≥98.0%
Identification (FTIR)Conforms
Refractive Index1.4890-1.4940 @ 20°C
3-Chloropropanesulfonyl Chloride is an intermediate used in the preparation of many enzymatic inhibitors. It is also used in the generation and trapping of the derived sulfene with imines and glyoxylates in the presence of chinchona alkaloids provide chiral sultams and sultones. It is mainly used for the synthesis of pharmaceutical intermediates.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Chloropropanesulfonyl Chloride is an intermediate used in the preparation of many enzymatic inhibitors. It is also used in the generation and trapping of the derived sulfene with imines and glyoxylates in the presence of chinchona alkaloids provide chiral sultams and sultones. It is mainly used for the synthesis of pharmaceutical intermediates.
Solubility
Soluble in dichloromethane, ethyl Acetate and hexane.
Notes
Store at -20°C. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Containers which are opened must be carefully resealed and kept upright to prevent leakage. Moisture sensitive. Keep away from alcohols, strong bases, oxidizing agents.
3-Chloropropanesulfonyl Chloride is an intermediate used in the preparation of many enzymatic inhibitors. It is also used in the generation and trapping of the derived sulfene with imines and glyoxylates in the presence of chinchona alkaloids provide chiral sultams and sultones. It is mainly used for the synthesis of pharmaceutical intermediates.
Solubility
Soluble in dichloromethane, ethyl Acetate and hexane.
Notes
Store at -20°C. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Containers which are opened must be carefully resealed and kept upright to prevent leakage. Moisture sensitive. Keep away from alcohols, strong bases, oxidizing agents.
RUO – Research Use Only
General References:
- RK Bakshi.; Q Hong.; R Tang.; RN Kalyani. Optimization of a privileged structure leading to potent and selective human melanocortin subtype-4 receptor ligands. span Bioorganic & Medicinal Chemistry Letters. 2006 16, (5) , 1130-1133.
- C Shah.; L McAtee.; JG Breitenbucher.; D Rudolph. Novel human histamine H 3 receptor antagonists. Bioorganic & Medicinal Chemistry Letters. 2002 12, (22) , 3309-3312.