Search
Thermo Scientific Chemicals
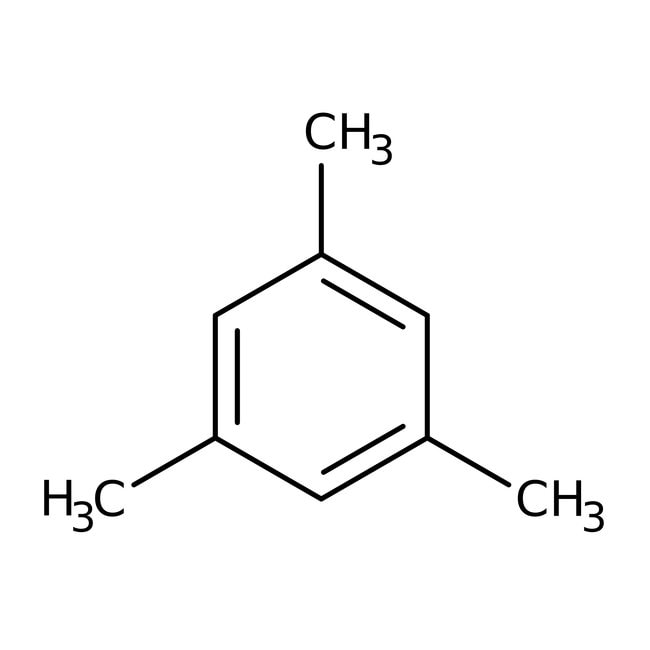
Mesitylene, 98+%
CAS: 108-67-8 | C9H12 | 120.195 g/mol
化学物質識別子
CAS108-67-8
IUPAC Name1,3,5-trimethylbenzene
Molecular FormulaC9H12
InChI KeyAUHZEENZYGFFBQ-UHFFFAOYSA-N
SMILESCC1=CC(C)=CC(C)=C1
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear colorless to very pale yellow
FormLiquid
Identification (FTIR)Conforms
Assay (GC)≥98.0%
Refractive Index1.4970-1.5010 @ 20?C
Mesitylene is used to make plastics and dyes. It acts as a solvent, ligand in organometallic chemistry and precursor to 2,4,6-trimethylaniline. It is also used as a developer for photopatternable silicones due to its solvent properties in the electronics industry. It is used as an internal standard in nuclear magnetic resonance (NMR) samples due to the presence of three equivalent protons in it. It is involved in the production of trimesic acid and antioxygen, epoxy firming agent and polyester resin stabilizers. Further, it is used as an additive and component in aviation gasoline blends.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Mesitylene is used to make plastics and dyes. It acts as a solvent, ligand in organometallic chemistry and precursor to 2,4,6-trimethylaniline. It is also used as a developer for photopatternable silicones due to its solvent properties in the electronics industry. It is used as an internal standard in nuclear magnetic resonance (NMR) samples due to the presence of three equivalent protons in it. It is involved in the production of trimesic acid and antioxygen, epoxy firming agent and polyester resin stabilizers. Further, it is used as an additive and component in aviation gasoline blends.
Solubility
Partly miscible with water.
Notes
Incompatible with strong oxidizing agents.
Mesitylene is used to make plastics and dyes. It acts as a solvent, ligand in organometallic chemistry and precursor to 2,4,6-trimethylaniline. It is also used as a developer for photopatternable silicones due to its solvent properties in the electronics industry. It is used as an internal standard in nuclear magnetic resonance (NMR) samples due to the presence of three equivalent protons in it. It is involved in the production of trimesic acid and antioxygen, epoxy firming agent and polyester resin stabilizers. Further, it is used as an additive and component in aviation gasoline blends.
Solubility
Partly miscible with water.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- The Adams modification of the Gattermann reaction for aromatic formylation by reaction with Zinc cyanide, L15836 , and HCl is exemplified: Org. Synth. Coll., 3, 549 (1955). The Vilsmeier formylation (see N,N-Dimethyl formamide, A13547 ), has been extended to less activated substrates, including mesitylene, by use of the combination of DMF and triflic anhydride: J. Chem. Soc., Chem. Commun., 1571 (1990).
- For conversion to mesitoic acid by Friedel-Crafts reaction with oxalyl chloride, and aqueous work-up, see: Org. Synth. Coll., 5, 706 (1973).
- Singh, P. Synthesis of mesitylene-based polyamine dendrimer for functionalisation of single-walled carbon nanotubes. J. Exp. Nanosci. 2015, 10 (6), 429-437.
- Medina, D. D.; Rotter, J. M.; Hu, Y.; Dogru, M.; Werner, V.; Auras, F.; Markiewicz, J. T.; Knochel, P.; Bein, T. Room Temperature Synthesis of Covalent-Organic Framework Films through Vapor-Assisted Conversion. J. Am. Chem. Soc. 2015, 137 (3), 1016-1019.