Search
Thermo Scientific Chemicals
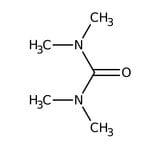
Tetramethylurea, 99%
CAS: 632-22-4 | C5H12N2O | 116.164 g/mol
化学物質識別子
CAS632-22-4
IUPAC Name1,1,3,3-tetramethylurea
Molecular FormulaC5H12N2O
InChI KeyAVQQQNCBBIEMEU-UHFFFAOYSA-N
SMILESCN(C)C(=O)N(C)C
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear colorless to pale yellow
FormLiquid
Identification (FTIR)Conforms
Assay (GC)≥98.5%
Refractive Index1.4500-1.4530 @ 20?C
Tetramethylurea is used as a solvent in dyestuff industries, in condensation reaction and intermediates in surfactant. It is utilized for base catalyzed isomerization and alkylation hydrocyanation due to its low permittivity. It reacts with oxalyl chloride to prepare tetramethyl chloroformamidinium chloride, which is used for the conversion of carboxylic acids and dialkyl phosphates to anhydrides and pyrophosphates respectively.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Tetramethylurea is used as a solvent in dyestuff industries, in condensation reaction and intermediates in surfactant. It is utilized for base catalyzed isomerization and alkylation hydrocyanation due to its low permittivity. It reacts with oxalyl chloride to prepare tetramethyl chloroformamidinium chloride, which is used for the conversion of carboxylic acids and dialkyl phosphates to anhydrides and pyrophosphates respectively.
Solubility
Miscible with water.Miscible with water, petroleum ether and common solvents.
Notes
Hygroscopic. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents.
Tetramethylurea is used as a solvent in dyestuff industries, in condensation reaction and intermediates in surfactant. It is utilized for base catalyzed isomerization and alkylation hydrocyanation due to its low permittivity. It reacts with oxalyl chloride to prepare tetramethyl chloroformamidinium chloride, which is used for the conversion of carboxylic acids and dialkyl phosphates to anhydrides and pyrophosphates respectively.
Solubility
Miscible with water.Miscible with water, petroleum ether and common solvents.
Notes
Hygroscopic. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- High-boiling dipolar aprotic solvent. The polar nature, wide liquid range and high solvent power of tetraalkylureas make them suitable solvents for a wide range of reactions; review: Angew. Chem. Int. Ed., 18, 503 (1979).
- Reacts with oxalyl chloride to give tetramethyl chloroformamidinium chloride, a useful reagent for the conversion of carboxylic acids and dialkyl phosphates to anhydrides and pyrophosphates respectively: Bull. Chem. Soc. Jpn., 56, 3529 (1983).
- Indra, S.; Biswas, R. Hydrogen-bond dynamics of water in presence of an amphiphile, tetramethylurea: signature of confinement-induced effects. Mol. Simul. 2015, 41 (5-6), 471-482.
- Krakowiak, J.; Wawer, J. Effect of temperature and ionic strength on volumetric and acoustic properties of solutions of urea alkyl derivatives in aqueous NaCl. J. Chem. Thermodyn. 2015, 90, 232-241.