Search
Thermo Scientific Chemicals
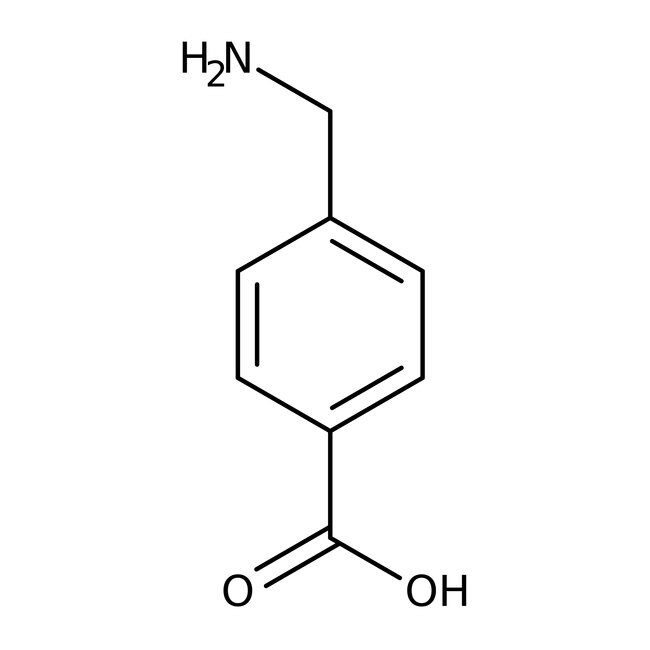
4-(Aminomethyl)benzoic acid, 97%
CAS: 56-91-7 | C8H9NO2 | 151.165 g/mol
化学物質識別子
CAS56-91-7
IUPAC Name4-(aminomethyl)benzoic acid
Molecular FormulaC8H9NO2
InChI KeyQCTBMLYLENLHLA-UHFFFAOYSA-N
SMILESNCC1=CC=C(C=C1)C(O)=O
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White to cream
Assay (HPLC)>96.0%
Water Content (Karl Fischer Titration)≤4.0%
FormPowder
Assay (Non-aqueous acid-base Titration)>96.0% (dry wt. basis)
4-(Aminomethyl)benzoic acid acts as an unnatural amino acid derivative. It is also used as a type 2 antifibrinolytic agent. Further, it reacts with 2-methyl-isothiourea sulfate to prepare 4-guanidinomethylbenzoic acid.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-(Aminomethyl)benzoic acid acts as an unnatural amino acid derivative. It is also used as a type 2 antifibrinolytic agent. Further, it reacts with 2-methyl-isothiourea sulfate to prepare 4-guanidinomethylbenzoic acid.
Solubility
Slightly soluble in water. Insoluble in ethanol, benzene and chloroform.
Notes
Incompatible with strong oxidizing agents, strong reducing agents and strong bases.
4-(Aminomethyl)benzoic acid acts as an unnatural amino acid derivative. It is also used as a type 2 antifibrinolytic agent. Further, it reacts with 2-methyl-isothiourea sulfate to prepare 4-guanidinomethylbenzoic acid.
Solubility
Slightly soluble in water. Insoluble in ethanol, benzene and chloroform.
Notes
Incompatible with strong oxidizing agents, strong reducing agents and strong bases.
RUO – Research Use Only
General References:
- Li, Z.; Fang, L.; Wang, J.; Dong, L.; Guo, Y.; Xie, Y. An Improved and Practical Synthesis of Tranexamic Acid. Org. Process Res. Dev. 2015, 19 (3), 444-448.
- Li, K. S.; Xiao, P.; Zhang, D. I.; Hou, X. B.; Ge, L.; Yang, D. X.; Liu, H. D.; He, D. F.; Chen, X.; Han, K. R.; Song, X. Y.; Yu, X.; Fang, H.; Sun, J. P. Identification of para-Substituted Benzoic Acid Derivatives as Potent Inhibitors of the Protein Phosphatase Slingshot. ChemMedChem 2015, 10 (12), 1980-1987.