Search
Thermo Scientific Chemicals
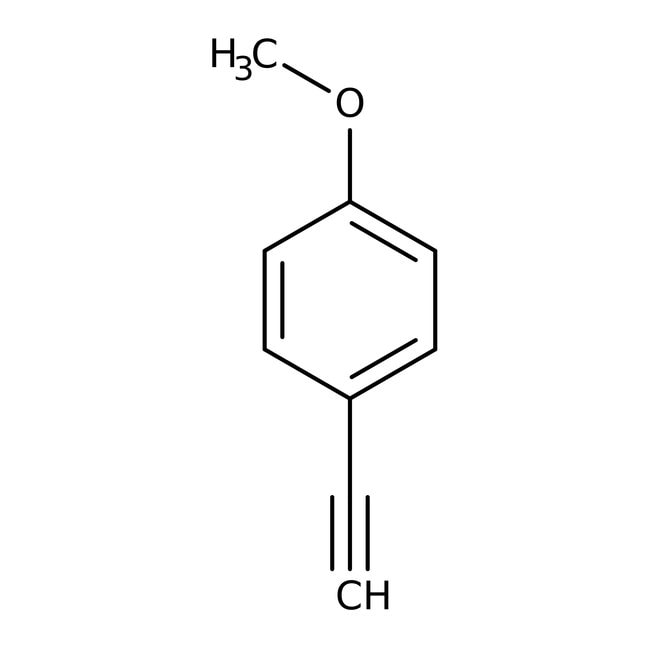
4-Methoxyphenylacetylene, 98%
CAS: 768-60-5 | C9H8O | 132.162 g/mol
化学物質識別子
CAS768-60-5
IUPAC Name1-ethynyl-4-methoxybenzene
Molecular FormulaC9H8O
InChI KeyKBIAVTUACPKPFJ-UHFFFAOYSA-N
SMILESCOC1=CC=C(C=C1)C#C
さらに表示
仕様 スペックシート
スペックシート
FormLiquid
Appearance (Color)Clear colorless to yellow
Assay (GC)≥98.5%
Refractive Index1.5640-1.5670 @ 20?C
Identification (FTIR)Conforms
4-Ethynylanisole was used in the synthesis of photo luminescent 1,2-dihydrophosphinines via a [4 + 2] cycloaddition. Along with an arylboronic acid and sodium azide in a copper-catalyzed, three-component synthesis of trisubstituted 1,2,4-triazoles. In a study of a gold (III)-catalyzed hydroamination of alkynes leading to N-vinylindoles3.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Ethynylanisole was used in the synthesis of photo luminescent 1,2-dihydrophosphinines via a [4 + 2] cycloaddition. Along with an arylboronic acid and sodium azide in a copper-catalyzed, three-component synthesis of trisubstituted 1,2,4-triazoles. In a study of a gold (III)-catalyzed hydroamination of alkynes leading to N-vinylindoles3.
Solubility
Insoluble in water. Soluble in chloroform, acetone, dichloromethane, and methanol.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
4-Ethynylanisole was used in the synthesis of photo luminescent 1,2-dihydrophosphinines via a [4 + 2] cycloaddition. Along with an arylboronic acid and sodium azide in a copper-catalyzed, three-component synthesis of trisubstituted 1,2,4-triazoles. In a study of a gold (III)-catalyzed hydroamination of alkynes leading to N-vinylindoles3.
Solubility
Insoluble in water. Soluble in chloroform, acetone, dichloromethane, and methanol.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Laura C Pavelka,; Kim M Baines.Facile synthesis of luminescent benzo-1,2-dihydrophosphinines from a phosphaalkene.. Dalton Transactions. 2012, 41 11,3294-3301.
- Yuhua Zhang et. al. Gold(III)-catalyzed double hydroamination of o-alkynylaniline with terminal alkynes leading to N-vinylindoles. Organic Letters. 2007 , 9 (4), 627-630.