Search
Thermo Scientific Chemicals
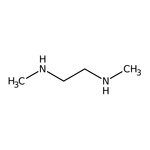
N,N'-Dimethylethylenediamine, 95%
CAS: 110-70-3 | C4H12N2 | 88.154 g/mol
| 製品番号(カタログ番号) | 数量 |
|---|---|
| L02204.14 または、製品番号L02204-14 | 25 g |
化学物質識別子
CAS110-70-3
IUPAC Namemethyl[2-(methylamino)ethyl]amine
Molecular FormulaC4H12N2
InChI KeyKVKFRMCSXWQSNT-UHFFFAOYSA-N
SMILESCNCCNC
さらに表示
仕様 スペックシート
スペックシート
Refractive Index1.4270-1.4330 @ 20?C
FormLiquid
Identification (FTIR)Conforms
Appearance (Color)Clear colorless to pale yellow
Assay (GC)≥94.0%
N,N'-Dimethylethylenediamine is used in DNA binding effect. It is used to enhance the adsorption of carbon dioxide. It acts as a ligand and form coordination complex such as dinitrato(N,N'-dimethyl-1,2-ethanediamine)copper(II) and dichloro(1,4-bis-(diphenyl phosphino)butane)-(1,2-ethylenediamine)ruthenium(II).
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
N,N′-Dimethylethylenediamine is used in DNA binding effect. It is used to enhance the adsorption of carbon dioxide. It acts as a ligand and form coordination complex such as dinitrato(N,N′-dimethyl-1,2-ethanediamine)copper(II) and dichloro(1,4-bis-(diphenyl phosphino)butane)-(1,2-ethylenediamine)ruthenium(II).
Solubility
Miscible with chloroform and dichloromethane.
Notes
Incompatible with acids, acid chlorides, acid anhydrides, strong oxidizing agents and carbon dioxide.
N,N′-Dimethylethylenediamine is used in DNA binding effect. It is used to enhance the adsorption of carbon dioxide. It acts as a ligand and form coordination complex such as dinitrato(N,N′-dimethyl-1,2-ethanediamine)copper(II) and dichloro(1,4-bis-(diphenyl phosphino)butane)-(1,2-ethylenediamine)ruthenium(II).
Solubility
Miscible with chloroform and dichloromethane.
Notes
Incompatible with acids, acid chlorides, acid anhydrides, strong oxidizing agents and carbon dioxide.
RUO – Research Use Only
General References:
- Reagent for protection of aldehyde groups as 2-substituted-1,3-dimethylimidazolidines, e.g. by azeotropic dehydration with benzene. The derivatives are stable to n-BuLi and LDA, and can be cleaved by acid hydrolysis, or in high yield by quaternization with MeI and mild hydrolysis: Tetrahedron, 41, 3803 (1985). With aromatic aldehydes, the imidazolidine derivatives are activated to ortho-lithiation, providing a route to o-substituted benzaldehydes: J. Org. Chem., 44, 2004 (1979).
- Kürkçüoğlu, G. S.; Yeşilel, O. Z.; Aksel, M.; Büyükgüngör, O. Hetero-octanuclear cubane-like and one-dimensional cyano complexes with the N,N-dimethylethylenediamine ligand. Polyhedron 2015, 85, 720-726.
- Mandal, S.; Naskar, B.; Modak, R.; Sikdar, Y.; Chatterjee, S.; Biswas, S.; Mondal, T. K.; Modak, D.; Goswami, S. Syntheses, crystal structures, spectral study and DFT calculation of three new copper(II) complexes derived from pyridoxal hydrochloride, N,N-dimethylethylenediamine and N,N-diethylethylenediamine. J. Mol. Struct. 2015, 1088, 38-49.