Search
Thermo Scientific Chemicals
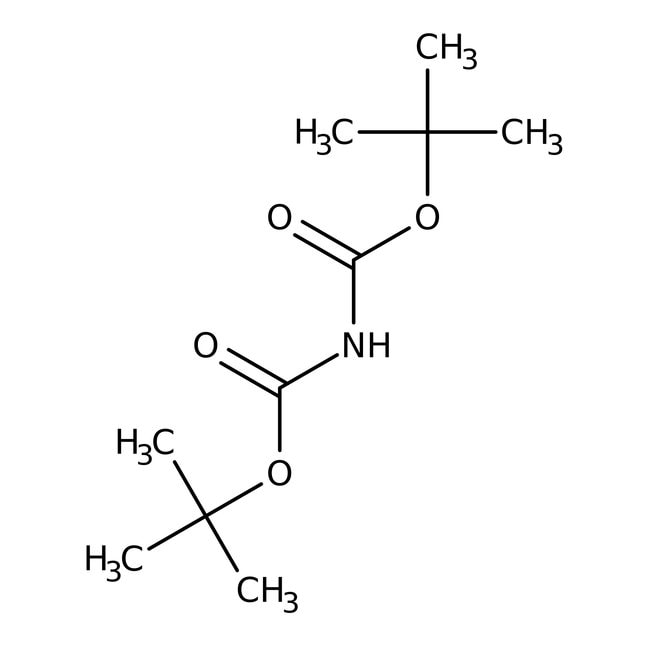
Di-tert-butyl iminodicarboxylate, 98%
CAS: 51779-32-9 | C10H19NO4 | 217.27 g/mol
化学物質識別子
CAS51779-32-9
IUPAC Nametert-butyl N-[(tert-butoxy)carbonyl]carbamate
Molecular FormulaC10H19NO4
InChI KeyXCAQIUOFDMREBA-UHFFFAOYSA-N
SMILESCC(C)(C)OC(=O)NC(=O)OC(C)(C)C
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White to cream
FormCrystals or powder or crystalline powder
Assay (GC)≥97.5%
Di-tert-butyl-iminodicarboxylate may be used in the preparation of trans-diamino-2-butene and cis-1,4-diamino-2-butene; di-tert-butyl N-3-butenyliminodicarboxylate; N,N-di-tert-butyl[(2-fluoro-4-nitro)benzylamino]dicarboxylate & C1-C20 and C21-C40 fragments of tetrafibricin. Modified Gabriel Reagent.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Di-tert-butyl-iminodicarboxylate may be used in the preparation of trans-diamino-2-butene and cis-1,4-diamino-2-butene; di-tert-butyl N-3-butenyliminodicarboxylate; N,N-di-tert-butyl[(2-fluoro-4-nitro)benzylamino]dicarboxylate & C1-C20 and C21-C40 fragments of tetrafibricin. Modified Gabriel Reagent.
Solubility
Insoluble in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents and air.
Di-tert-butyl-iminodicarboxylate may be used in the preparation of trans-diamino-2-butene and cis-1,4-diamino-2-butene; di-tert-butyl N-3-butenyliminodicarboxylate; N,N-di-tert-butyl[(2-fluoro-4-nitro)benzylamino]dicarboxylate & C1-C20 and C21-C40 fragments of tetrafibricin. Modified Gabriel Reagent.
Solubility
Insoluble in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents and air.
RUO – Research Use Only
General References:
- K Kashiwagi,; A Kuraishi,; H Tomitori,; A Igarashi,; K Nishimura,; A Shirahata, K Igarashi.Identification of the putrescine recognition site on polyamine transport protein PotE.. Mikrobiologiya. 2000, 27536007-36012.
- Young Ah Kim,; Ashoke Sharon,; Chung K Chu,; Reem H Rais,; Omar N Al Safarjalani,; Fardos N M Naguib,; Mahmoud H el Kouni. Synthesis, biological evaluation and molecular modeling studies of N6-benzyladenosine analogues as potential anti-toxoplasma agents. Biochemical Pharmacology. 2011 , 73 (10),1558-1572.
- Gudipati V,; Curran DP. Synthesis of C1-C20 and C21-C40 fragments of tetrafibricin.. Journal name. 2011 , 52 (17), 2254-2257.