Search
Thermo Scientific Chemicals
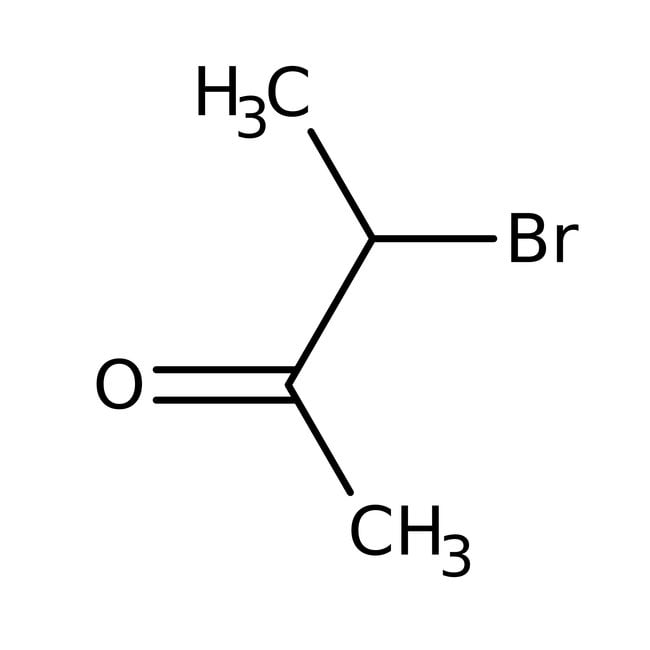
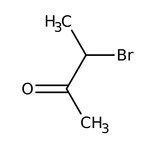
3-Bromo-2-butanone, 97%, stab. with <1% magnesium oxide
CAS: 814-75-5 | C4H7BrO | 151.003 g/mol
化学物質識別子
CAS814-75-5
IUPAC Name3-bromobutan-2-one
Molecular FormulaC4H7BrO
InChI KeyBNBOUFHCTIFWHN-UHFFFAOYNA-N
SMILESCC(Br)C(C)=O
さらに表示
仕様 スペックシート
スペックシート
FormLiquid
CommentMay contain small amount of sediment due to MgO stabilizer.
Appearance (Color)Clear, colorless to yellow
Assay (GC)>96.0%
Refractive Index1.4565 - 1.4615 @ 20?C
3-Bromo-2-butanone is involved in the preparation of o-isopropyl S-3-oxobutan-2-yl dithiocarbonate by reaction with potassium o-isopropylxanthate, which on treatment with sulfuric acid gives 4,5-dimethyl-1,3-dithiol-2-one .
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Bromo-2-butanone is involved in the preparation of o-isopropyl S-3-oxobutan-2-yl dithiocarbonate by reaction with potassium o-isopropylxanthate, which on treatment with sulfuric acid gives 4,5-dimethyl-1,3-dithiol-2-one .
Solubility
Miscible with dichloromethane.
Notes
Incompatible with strong oxidizing agents, strong bases and strong reducing agents.
3-Bromo-2-butanone is involved in the preparation of o-isopropyl S-3-oxobutan-2-yl dithiocarbonate by reaction with potassium o-isopropylxanthate, which on treatment with sulfuric acid gives 4,5-dimethyl-1,3-dithiol-2-one .
Solubility
Miscible with dichloromethane.
Notes
Incompatible with strong oxidizing agents, strong bases and strong reducing agents.
RUO – Research Use Only
General References:
- Has been used in an improved synthesis of 5-halopyrimidines by the reaction of ɑ -halo ketones with nitriles: Synthesis, 1053 (1992).
- Goel, R.; Luxami, V.; Paul, K. Recent advances in development of imidazo[1,2-a]pyrazines: synthesis, reactivity and their biological applications. Org. Biomol. Chem. 2015, 13 (12), 3525-3555.
- Ghosh, A. C.; Reinhardt, J. K.; Kindermann, M. K.; Schulzke, C. The ring opening reaction of 1,3-dithiol-2-one systems is fully reversible. Chem. Commun. 2014, 50 (70), 10102-10104.