Search
Thermo Scientific Chemicals
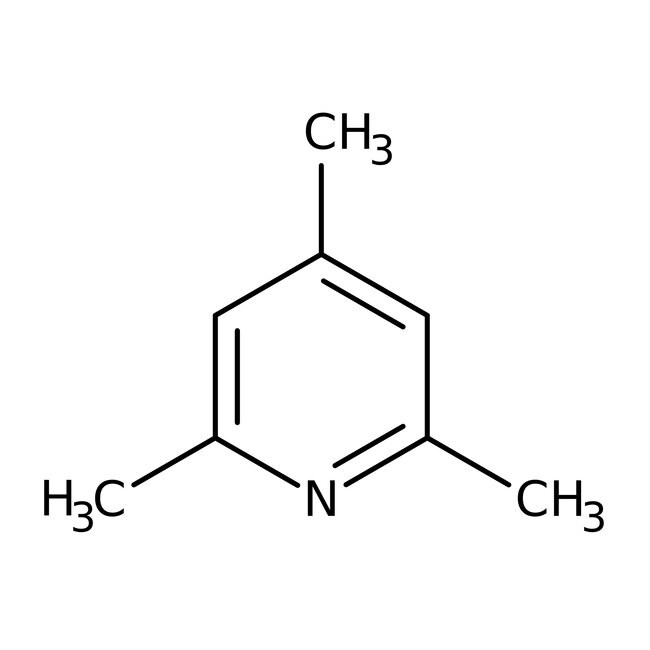
2,4,6-Collidine, 99%
CAS: 108-75-8 | C8H11N | 121.18 g/mol
化学物質識別子
CAS108-75-8
IUPAC Name2,4,6-trimethylpyridine
Molecular FormulaC8H11N
InChI KeyBWZVCCNYKMEVEX-UHFFFAOYSA-N
SMILESCC1=CC(C)=NC(C)=C1
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear, colorless to yellow
Assay (GC)≥98.5%
Identification (FTIR)Conforms
CommentMay darken on storage
Refractive Index1.4965-1.5005 @ 20?C
さらに表示
2,4,6-Collidine is used as a tissue fixative for electron microscopy. It is useful in dehydrohalogenation reactions and acts as a solvent for the cleavage of hindered esters by anhydrous lithium iodide.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2,4,6-Collidine is used as a tissue fixative for electron microscopy. It is useful in dehydrohalogenation reactions and acts as a solvent for the cleavage of hindered esters by anhydrous lithium iodide.
Solubility
Slightly miscible with water. Miscible with ether, methanol, acetone, chloroform, benzene, toluene and dilute acids.
Notes
Incompatible with oxidizing agents and strong acids.
2,4,6-Collidine is used as a tissue fixative for electron microscopy. It is useful in dehydrohalogenation reactions and acts as a solvent for the cleavage of hindered esters by anhydrous lithium iodide.
Solubility
Slightly miscible with water. Miscible with ether, methanol, acetone, chloroform, benzene, toluene and dilute acids.
Notes
Incompatible with oxidizing agents and strong acids.
RUO – Research Use Only
General References:
- Hindered base. For use in the dehydrochlorination of ɑ-chloroketones to enones, see: Org. Synth. Coll., 4, 162 (1963). For use in combination with Trifluoromethanesulfonic anhydride, A11767 for the generation of ketenimines from olefins, see N,N-Dimethyl acetamide, A10924.
- Superior base for peptide coupling reactions, causing less racemization than the more commonly used N-ethyldiisopropylamine or N-methylmorpholine: J. Org. Chem., 59, 695 (1994). For comparison with other hindered bases, see: J. Org. Chem., 61, 2460 (1996). For peptide reagents, see Appendix 6.
- Solvent for various O-alkyl cleavage reaction by LiI, including hindered esters: J. Org. Chem., 28, 2184 (1963), and methyl ethers of phenols: Chem. Commun., 616 (1969).
- Dufour-Gallant, J.; Chatenet, D.; Lubell, W. D. De Novo Conception of Small Molecule Modulators Based on Endogenous Peptide Ligands: Pyrrolodiazepin-2-one γ-Turn Mimics That Differentially Modulate Urotensin II Receptor-Mediated Vasoconstriction ex Vivo. J. Med. Chem. 2015, 58 (11), 4624-4637.
- Fujioka, H.; Matsumoto, N.; Ohta, R.; Yamakawa, M.; Shimizu, N.; Kimura, T.; Murai, K. Organic synthesis based on the Beckmann fragmentation: generation of an electrophilic salt intermediate and successive C-C bond formation using Gilman reagents. Tetrahedron Lett. 2015, 56 (21), 2656-2658.