Search
Thermo Scientific Chemicals
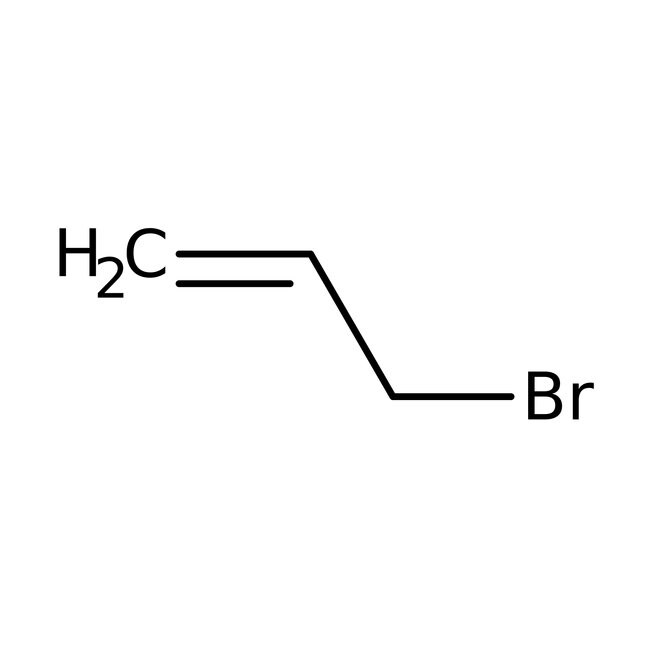
Allyl bromide, 99%, stab. with 300-1000ppm Propylene oxide
CAS: 106-95-6 | C3H5Br | 120.977 g/mol
化学物質識別子
CAS106-95-6
IUPAC Name3-bromoprop-1-ene
Molecular FormulaC3H5Br
InChI KeyBHELZAPQIKSEDF-UHFFFAOYSA-N
SMILESBrCC=C
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)Clear colorless to pale yellow
FormLiquid
Assay (GC)≥98.5%
Refractive Index1.4670-1.4710 @ 20°C
StabilizerPropylene oxide, 300-1,000ppm
Allyl bromide is used as an alkylating agent in the synthesis of pharmaceuticals, polymers, adhesives, perfumes, biochemicals and other allylic compounds. It is used as precursor for the preparation of allyliczinc bromide by reacting it with zinc. It is also used in the preparation of allylethers like allyl decyl ether, allyl benzyl ether and allyl geranyl ether. It is also used in the preparation of R enantiomer of allyl phenyl carbinol (APC) such as 1-phenyl-3-butene, which is a valuable intermediate for drugs and agro-chemicals.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Allyl bromide is used as an alkylating agent in the synthesis of pharmaceuticals, polymers, adhesives, perfumes, biochemicals and other allylic compounds. It is used as precursor for the preparation of allyliczinc bromide by reacting it with zinc. It is also used in the preparation of allylethers like allyl decyl ether, allyl benzyl ether and allyl geranyl ether. It is also used in the preparation of R enantiomer of allyl phenyl carbinol (APC) such as 1-phenyl-3-butene, which is a valuable intermediate for drugs and agro-chemicals.
Solubility
Slightly miscible with water.
Notes
Moisture and light sensitive. Incompatible with oxidizing agents, alkali metals, alkaline earth metals, amides, amines and powdered metals.
Allyl bromide is used as an alkylating agent in the synthesis of pharmaceuticals, polymers, adhesives, perfumes, biochemicals and other allylic compounds. It is used as precursor for the preparation of allyliczinc bromide by reacting it with zinc. It is also used in the preparation of allylethers like allyl decyl ether, allyl benzyl ether and allyl geranyl ether. It is also used in the preparation of R enantiomer of allyl phenyl carbinol (APC) such as 1-phenyl-3-butene, which is a valuable intermediate for drugs and agro-chemicals.
Solubility
Slightly miscible with water.
Notes
Moisture and light sensitive. Incompatible with oxidizing agents, alkali metals, alkaline earth metals, amides, amines and powdered metals.
RUO – Research Use Only
General References:
- Pandey, G.; Kant, R.; Batra, S. A general route to the synthesis of indoloazocines using allyl bromides prepared from Morita-Baylis-Hillman adducts. Tetrahedron Lett. 2015, 56 (7), 930-933.
- Fananas-Mastral, M.; Vitale, R.; Perez, M.; Feringa, B. L. Enantioselective Synthesis of All-Carbon Quaternary Stereogenic Centers via Copper-Catalyzed Asymmetric Allylic Alkylation of (Z)-Allyl Bromides with Organolithium Reagents. Chem. Eur. J. 2015, 21 (11), 4209-4212.