Search
Thermo Scientific Chemicals
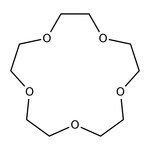
15-Crown-5, 98%
CAS: 33100-27-5 | C10H20O5 | 220.265 g/mol
化学物質識別子
CAS33100-27-5
IUPAC Name1,4,7,10,13-pentaoxacyclopentadecane
Molecular FormulaC10H20O5
InChI KeyVFTFKUDGYRBSAL-UHFFFAOYSA-N
SMILESC1COCCOCCOCCOCCO1
さらに表示
仕様 スペックシート
スペックシート
Assay (GC)≥97.5%
Appearance (Color)Clear colorless to pale yellow
Water Content (Karl Fischer Titration)≤1%
Identification (FTIR)Conforms
Refractive Index1.4640-1.4680 @ 20?C
さらに表示
15-Crown-5 is used as an efficient phase transfer catalyst and as a complexing agent. It is used to isolate oxonium ion (H7O3)+ salts especially from a solution of tetrchloroauric acid. It catalyzes the O-alkylation of the sodium salts of carboxylic acids in the penicillin and cephalosporin series, thereby facilitating esterification reaction without usage of acid. It is also used to facilitate the Williamson synthesis of ethers with hindered alcohols and sodium hydride. It is used with lithium aluminum hydride, for performing reduction reactions in hydrocarbon solvents. Further, it is involved in the Horner-Wadsworth-Emmons reaction to prepare stilbenes from aldehydes.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
15-Crown-5 is used as an efficient phase transfer catalyst and as a complexing agent. It is used to isolate oxonium ion (H7O3)+ salts especially from a solution of tetrchloroauric acid. It catalyzes the O-alkylation of the sodium salts of carboxylic acids in the penicillin and cephalosporin series, thereby facilitating esterification reaction without usage of acid. It is also used to facilitate the Williamson synthesis of ethers with hindered alcohols and sodium hydride. It is used with lithium aluminum hydride, for performing reduction reactions in hydrocarbon solvents. Further, it is involved in the Horner-Wadsworth-Emmons reaction to prepare stilbenes from aldehydes.
Solubility
Miscible with organic solvents.
Notes
Hygroscopic. Moisture sensitive. Incompatible with strong oxidizing agents and strong acids.
15-Crown-5 is used as an efficient phase transfer catalyst and as a complexing agent. It is used to isolate oxonium ion (H7O3)+ salts especially from a solution of tetrchloroauric acid. It catalyzes the O-alkylation of the sodium salts of carboxylic acids in the penicillin and cephalosporin series, thereby facilitating esterification reaction without usage of acid. It is also used to facilitate the Williamson synthesis of ethers with hindered alcohols and sodium hydride. It is used with lithium aluminum hydride, for performing reduction reactions in hydrocarbon solvents. Further, it is involved in the Horner-Wadsworth-Emmons reaction to prepare stilbenes from aldehydes.
Solubility
Miscible with organic solvents.
Notes
Hygroscopic. Moisture sensitive. Incompatible with strong oxidizing agents and strong acids.
RUO – Research Use Only
General References:
- Crown ether (see Appendix 2) especially suited to complexing sodium ions. Thus, catalyzes the O-alkylation of the Na salts of carboxylic acids in the penicillin and cephalosporin series, providing a method for esterification without exposure to acid: Synthesis, 52, (1986).
- Greatly facilitates the Williamson synthesis of ethers with hindered alcohols and NaH: Tetrahedron Lett., 38, 4679 (1997). Promotes the cleavage of unsymmetrical ethers by Sodium iodide, A15480, in the opposite sense to BBr3, i.e. by nucleophilic displacement from the less substituted carbon atom: Tetrahedron Lett., 22, 4239 (1981).
- Greatly increases the reactivity of phosphonate anions generated by reaction with NaH in the Horner-Wadsworth-Emmons reaction. Thus, has been used in a high-yield synthesis of stilbenes from aldehydes and Diethyl benzyl phosphonate, A10645: Synthesis, 117 (1981).
- Use in combination with Lithium aluminum hydride, A18116, enables reductions to be carried out in hydrocarbon solvents: J. Chem. Soc., Chem. Commun., 1234 (1985).
- Kuo, T. Y.; Tseng, W. H.; Chen, C. H. Force Spectroscopy of Metal-Crown Ether Multivalency: Effect of Local Environments on Energy Landscape and Sensing Kinetics. Angew. Chem. Int. Ed. 2015, 54 (32), 9213-9217.
- Inokuchi, Y.; Ebata, T.; Rizzo, T. R. Solvent Effects on the Encapsulation of Divalent Ions by Benzo-18-Crown-6 and Benzo-15-Crown-5. J. Phys. Chem. A 2015, 119 (29), 8097-8105.