Search
Thermo Scientific Chemicals
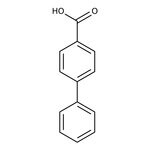
Biphenyl-4-carboxylic acid, 98%
CAS: 92-92-2 | C13H10O2 | 198.221 g/mol
化学物質識別子
CAS92-92-2
IUPAC Name[1,1'-biphenyl]-4-carboxylic acid
Molecular FormulaC13H10O2
InChI KeyNNJMFJSKMRYHSR-UHFFFAOYSA-N
SMILESOC(=O)C1=CC=C(C=C1)C1=CC=CC=C1
さらに表示
仕様 スペックシート
スペックシート
Appearance (Color)White to cream or pale yellow
Assay (Aqueous acid-base Titration)≥97.5 to ≤102.5%
Assay (Silylated GC)≥97.5%
FormPowder
Melting Point (clear melt)222.0-230.0?C
Biphenyl-4-carboxylic acid is employed in the synthesis, characterization of europium, terbium complexes.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Biphenyl-4-carboxylic acid is employed in the synthesis, characterization of europium, terbium complexes.
Solubility
Insoluble in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with bases and oxidizing agents.
Biphenyl-4-carboxylic acid is employed in the synthesis, characterization of europium, terbium complexes.
Solubility
Insoluble in water.
Notes
Keep container tightly sealed. Store in cool, dry conditions in well sealed containers. Incompatible with bases and oxidizing agents.
RUO – Research Use Only
General References:
- Franck Picard.; Tobias Schulz.; Rolf W. Hartmann. 5-Phenyl substituted 1-methyl-2-pyridones and 4'-substituted biphenyl-4-carboxylic acids. synthesis and evaluation as inhibitors of steroid-5α-reductase type 1 and 2. Bioorganic & Medicinal Chemistry. 2002, 10 (2), 437-448.
- Zhao Yongliang.; Zhao Fengying.; Li Qiang.; Gao Deqing. Synthesis, Characterization and Fluorescence Properties of Europium, Terbium Complexes with Biphenyl-4-Carboxylic Acid and o-Phenanthroline. Journal of Rare Earths. 2006, 24 (1), 18-22.